FDA Approves Lumisight (pegulicianine) Optical Imaging Agent to Illuminate Residual Breast Cancer Post-Lumpectomy
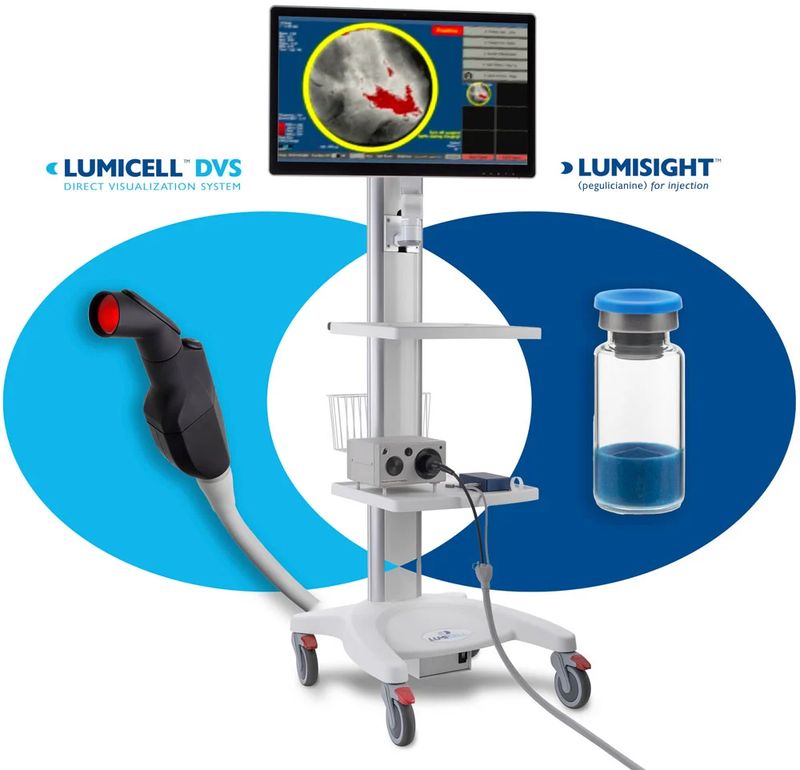
NEWTON, Mass.--(BUSINESS WIRE) April 18, 2024 -- Lumicell, Inc., a privately held company focused on developing innovative fluorescence-guided imaging technologies for cancerous tissue detection during surgery, today announced the U.S. Food & Drug Administration (FDA) approved the company’s New Drug Application (NDA) for its Lumisight™ (pegulicianine) optical imaging agent and its Premarket Approval (PMA) application for Lumicell™ Direct Visualization System (DVS), together referred to as LumiSystem™.
With 84% diagnostic accuracy, LumiSystem enables surgeons to scan the breast cavity post-lumpectomy, in real-time, to detect and resect residual cancer that may have otherwise been missed, potentially sparing some patients from second surgeries.1-2 The LumiSystem combination is indicated for fluorescence imaging in adults with breast cancer as an adjunct for the intraoperative detection of cancerous tissue within the resection cavity following removal of the primary specimen during lumpectomy surgery.
“We are immensely proud of the dual approval of Lumisight and Lumicell DVS – we believe this is the first drug-device combination product approved in over a decade to have followed both of the FDA's most stringent NDA and PMA review processes,”3 said Howard Hechler, President and Chief Operating Officer, Lumicell. “With the FDA’s approval, LumiSystem is now the first and only imaging combination product capable of detecting cancerous tissue where it matters most, inside the breast cavity.”
“Breast cancer is all too common, and sadly, 1 in 8 women will develop it during their lifetime,”4 said Kelly Hunt, MD, Chair of the Department of Breast Surgical Oncology at MD Anderson Cancer Center and President of the Society of Surgical Oncology. “Our most common surgical procedure to treat these women is lumpectomy. Unfortunately, the intraoperative tools we have are limited and do not identify the extent of tumor accurately enough, making it challenging to achieve a complete tumor resection, leading to as many as 36% of patients needing a second surgery.”5
“Up to 65% of the time, we do not find residual cancer in the second surgery and are left wondering if we performed an unnecessary surgery due to a false positive margin assessment or if the cancer was missed again in the second surgery,”6 said Irene Wapnir, MD, Breast Surgical Oncologist and Professor of Surgery, Stanford University School of Medicine.
“During lumpectomy surgery, surgeons still struggle to identify and remove all of the tumor during the first operation,”7 said Barbara Smith, MD, PhD, Director of the Breast Program at Massachusetts General Hospital and Professor of Surgery at Harvard Medical School. “With LumiSystem, we will now have a technology that is clinically proven to achieve a more complete cancer resection during lumpectomy that could help some patients avoid a second surgery.”2
The safety of the system was established using data from more than 700 breast cancer patients across five clinical studies at top academic and regional community cancer centers across the U.S. The most common side effects with Lumisight are hypersensitivity and an abnormal color in urine. Lumisight may cause serious hypersensitivity reactions, including anaphylaxis. Results of the Investigation of Novel Surgical Imaging for Tumor Excision (INSITE) pivotal trial, used to support the efficacy of the system, were published in NEJM Evidence.
“We want to thank our clinical investigators and the hundreds of women who participated in our breast program for Lumisight and Lumicell DVS,” said Jorge Ferrer, Chief Scientific Officer, Lumicell. “Due to your important contributions, Lumisight and Lumicell DVS are now approved and will be available in the United States shortly.”
Please visit www.LumiSystem.com to learn more about Lumisight and Lumicell DVS.
About Lumicell Inc.
Lumicell is a privately held company focused on enabling a more complete resection of cancer by advancing the development and commercialization of its innovative fluorescence guided surgery technology. The company’s lead products are Lumisight™ (pegulicianine) and Lumicell™ DVS which are designed for use in combination to illuminate cancerous tissue within the breast cavity during the initial lumpectomy procedure, as an adjunct to the Standard of Care. Lumicell’s proprietary, pan-oncologic optical imaging agent, Lumisight, is also being explored for further development across a wide variety of solid tumor indications. For more information, please visit www.Lumicell.com and follow the company on Facebook, X, and LinkedIn.
Indications for Use
Lumisight, an optical imaging agent, and Lumicell DVS, a fluorescence imaging device, are indicated for fluorescence imaging in adults with breast cancer as an adjunct for the intraoperative detection of cancerous tissue within the resection cavity following removal of the primary specimen during lumpectomy surgery.
Important Safety Information
What is the most important information I should know about Lumisight?
Lumisight may cause serious hypersensitivity reactions, including anaphylaxis. Anaphylaxis has occurred in 4/726 (0.6%) of patients in clinical studies. Tell your doctor if you have any history of hypersensitivity reactions to pegulicianine or to contrast media or products containing polyethylene glycol (PEG).
Your healthcare provider should have emergency resuscitation drugs, equipment, and trained personnel available during use of Lumisight. Healthcare providers should monitor all patients for hypersensitivity reactions and if one is suspected, immediately discontinue the injection and initiate appropriate therapy.
What other safety information should I know about Lumisight?
- Risk of Misdiagnosis: The absence of a signal in surgery does not rule out cancer. Additionally, a positive signal may be seen in non-cancerous tissue.
- Interference from Dyes Used for Sentinel Lymph Node Mapping: Your healthcare provider should avoid using dyes before imaging with Lumisight.
What are the most common side effects of Lumisight?
The most common side effects (≥1%) include hypersensitivity and an abnormal color in urine.
You are encouraged to report negative side effects of prescription drugs to the FDA. Visit MedWatch or call 1-800-FDA-1088. Please see full Prescribing Information, including Boxed Warning.
What is the most important information I should know about Lumicell DVS?
- Adjunctive use: Lumicell DVS is for use as part of the lumpectomy procedure and is not a replacement for the standard of care procedures and pathology. Your healthcare provider must be trained on proper use of the Lumicell DVS, and breast conserving surgery prior to performing any procedures.
- Risk of Misdiagnosis: The absence of a signal in surgery does not rule out cancer. Additionally, a positive signal may be seen in non-cancerous tissue.
- Interference from Dyes Used for Sentinel Lymph Node Mapping: Your healthcare provider should avoid using dyes before imaging with Lumisight.
- Tissue perforation or damage: During the procedure, your healthcare provider should avoid excessive pressure or torque with the handheld probe while inside the cavity as it can cause tissue perforation or damage.
- Infection risk: To avoid infection risk, the reusable handheld probe and cables should be properly and completely disinfected and reprocessed after each use. Your healthcare provider should also ensure proper use of the sterile, single-use probe cover.
- Eye discomfort: Avoid direct eye exposure to handheld light probe as it may cause pain.
- Diagnostic accuracy is based on true positive and true negative images detected in the LumiSystem pivotal trial
- Smith, et al., NEJM Evidence 2023; 2(7)
- Data on File – Lumicell
- American Cancer Society. Key Statistics for Breast Cancer. Accessed November 1, 2023
- Dupont, et al., Ann Surg 2021; 273(5): 876-881
- Bundred, et al., BMJ 2022; 378:e070346
- Tang, et.al., Am J Surg 2015
Source: Lumicell, Inc.
Posted: April 2024