New England Journal of Medicine Publishes Results of Ebola Clinical Trial Confirming Superiority of Regeneron's REGN-EB3 to ZMapp in Preventing Ebola Deaths
TARRYTOWN, N.Y., Nov. 27, 2019 /PRNewswire/ -- Regeneron Pharmaceuticals, Inc. (NASDAQ: REGN) announced today that the New England Journal of Medicine(NEJM) published results from the randomized controlled PALM trial showing that Regeneron's REGN-EB3 and another investigational agent provided the highest overall survival rates among four investigational treatments for Ebola virus disease. Monoclonal antibody treatments REGN-EB3, mAb114 and ZMapp and the small molecule antiviral agent remdesivir were given to a trial population of 681 patients who had Ebola during the ongoing outbreak in the Democratic Republic of the Congo (DRC). REGN-EB3, a triple-antibody cocktail, demonstrated superior efficacy compared to the ZMapp control arm across multiple measures, including the primary endpoint of mortality at day 28 (33.5 percent with REGN-EB3 versus 51.3 percent with ZMapp, p=0.002) and secondary endpoint of reduction of the number of days until the Ebola virus was no longer detected in the bloodstream.
The trial was stopped in August 2019 when preliminary results showed that REGN-EB3 crossed the pre-specified superiority threshold for preventing death compared to ZMapp. Although a second investigational treatment, mAb114, did not meet this statistical threshold, it had notable activity, and the decision was made for these two strongest-performing investigational therapies to continue to be administered to patients in a trial extension phase.
The REGN-EB3 antibodies were created using Regeneron's novel and proprietary VelocImmune® platform, which utilizes a mouse model with a genetically humanized immune system, and associated VelociSuite technologies that allow for harvesting and production of specific human antibodies from these mice. These technologies enable the rapid and efficient generation of multiple fully-human antibodies against targets such as Ebola virus, without requiring the identification and utilization of rare human survivors as sources of such therapeutic antibodies.
"In this study, REGN-EB3 demonstrated a significant survival advantage over another antibody treatment approach, underscoring the importance of antibody selection and design. REGN-EB3 was specifically designed to enhance efficacy, reduce the risk of viral resistance and mitigate against potential virus evolution. Our ability to rapidly create and develop these clinically-effective Ebola-specific antibodies highlights the power and speed of our VelociSuite human antibody platforms," said Neil Stahl, Ph.D., Executive Vice President of Research and Development at Regeneron. "We are committed to continuing to provide this important treatment to patients through the PALM trial extension trial and 'compassionate use' until the current outbreak ends. We are also working closely with the U.S. Food and Drug Administration to gain regulatory approval and with U.S. and global health authorities to determine appropriate stockpiling of REGN-EB3, as we believe our triple antibody approach may have particular utility for future outbreaks."
Ebola outbreaks have historically resulted in survival rates as low as 10 percent1 and there are no approved treatments for the disease. Of all patients given REGN-EB3 in this trial setting, 66.5 percent were alive at day 28, compared to 48.7 percent of patients given ZMapp. Importantly, administration of treatment earlier in the course of disease (when viral loads are typically lower) resulted in dramatically higher survival rates with all four investigational treatments, including 88.8 percent survival at day 28 with REGN-EB3. The greatest reduction in mortality (minus 22.5 percent) across all study groups occurred with REGN-EB3 treatment in patients who were treated later in the course of their disease, when risk of dying from Ebola is greatest.
All patients received optimized supportive care (oSOC), including supportive oral and/or intravenous fluids, electrolyte replacement, maintaining of oxygen status and blood pressure, pain management, and antibiotics and antimalarials as indicated. The paper reported 3 serious adverse events for REGN-EB3, compared to 7 for ZMapp, 9 for remdesivir and 10 for mAb114.
Comparison of 28-day mortality and days to negative PCR by treatment group
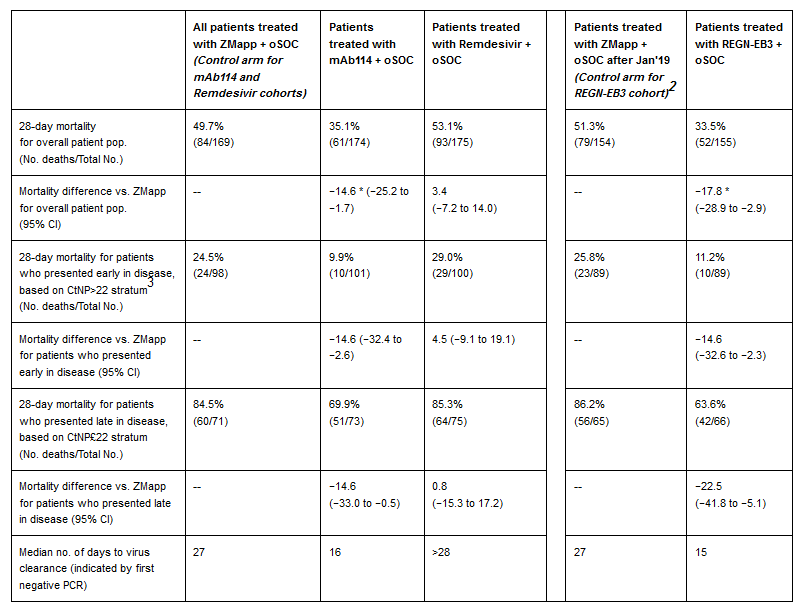
*Statistically significant according to interim monitoring boundary of p<0.034 and p<0.029 for mAb114 and REGN-EB3, respectively.
"This trial required an incredible level of cooperation from international government agencies, non-governmental organizations and drug developers in order to deliver experimental treatments to remote, violence-plagued areas of the DRC," said Sumathi Sivapalasingam, M.D., Senior Director, Early Clinical Development and Experimental Sciences at Regeneron. "Healthcare workers in the field risked their lives to treat patients, all while conducting scientifically rigorous and ethically sound research that will improve the treatment of Ebola in future outbreaks. We are so grateful for their work and that REGN-EB3 was able to improve survival for people suffering from this terrible disease."
About the PALM Trial
The PAmoja TuLinde Maisha (PALM ["together save lives" in Kiswahili]) clinical trial was a randomized, multicenter, controlled trial initiated in 2018 to evaluate the safety and efficacy of three investigational Ebola virus disease therapies: mAb114, remdesivir and ZMapp. The trial protocol was amended after the World Health Organization (WHO) held an Ad-Hoc Expert Consultation to assess all preclinical and clinical data on available investigational products, and recommended the addition of REGN-EB3 as a fourth treatment arm. The National Institutes of Health (NIH) and the Institut National de Recherche Biomédicale (INRB) in the DRC jointly sponsored and served as co-principal investigators and are senior authors of the New England Journal of Medicine publication.
About REGN-EB3
REGN-EB3 (also known as REGN3470-3471-3479) was invented by Regeneron using its VelociSuite® technologies, starting with the VelocImmune® mouse; the therapy combines three fully-human monoclonal antibodies. REGN-EB3 has received Orphan Drug designation from both the U.S. Food and Drug Administration (FDA) and the European Medicines Agency; in early September 2019, REGN-EB3 also received Breakthrough Therapy designation from the FDA. REGN-EB3 is being developed, tested and manufactured through contracts awarded in 2015 and 2017 by the Biomedical Advanced Research and Development Authority (BARDA), under the Assistant Secretary for Preparedness and Response within the U.S. Department of Health and Human Services (USG Contract No. HHSO100201500013C and HHSO100201700016C). REGN-EB3 is currently under clinical development and its safety and efficacy have not been fully evaluated by any regulatory authority.
Regeneron's rapid response infectious disease platform has the potential to accelerate response to future epidemics and pandemics that may pose a threat to public health. Regeneron and BARDA have several ongoing research collaborations in addition to the Ebola program, including efforts to develop antibodies targeting up to 10 pathogens that pose significant risk to public health. (Article from : www.drugs.com)

