Lilly and Dermira Present New Lebrikizumab Phase 2b Clinical Data Showing Patients with Atopic Dermatitis Had Clinically Meaningful Improvements in Itch, Sleep and Quality of Life Data presented at 2020 Annual Meeting
INDIANAPOLIS, June 12, 2020 /PRNewswire/ -- Eli Lilly and Company (NYSE: LLY) and Dermira, Inc., a wholly-owned subsidiary of Eli Lilly and Company, presented new data from the Phase 2b clinical trial of lebrikizumab in patients with moderate-to-severe atopic dermatitis. Data from this study suggests that treatment with lebrikizumab provided rapid and clinically meaningful improvements in itch, sleep and overall measures of quality of life.
Emma Guttman-Yassky, M.D., Ph.D., a leading investigator, presented the results of the data analysis during a virtual late-breaking, oral session at the American Academy of Dermatology (AAD) 2020 Annual Meeting.
"Understanding the potential for lebrikizumab to improve the skin symptoms of atopic dermatitis as well as other commonly associated symptoms, such as itch and loss of sleep, is critical to understanding its true potential to help patients," said Guttman-Yassky, the Sol and Clara Kest professor of dermatology, vice chair of the department of dermatology, director of the Center for Excellence in Eczema and director of the laboratory of inflammatory skin diseases in the department of dermatology at Icahn School of Medicine at Mount Sinai. "These results suggest that lebrikizumab could provide clinically meaningful improvement in treating symptoms that are most bothersome to patients."
The results of the data analysis showed that lebrikizumab improved symptoms and quality of life in a rapid, dose-dependent manner across a range of atopic dermatitis-specific and other measures compared with placebo. Specifically, lebrikizumab improved:
- Itch by Day 2 with further improvement to Week 16
- Sleep by the first on-treatment assessment at Week 1 with further improvement to Week 16
- Disease severity as assessed by the POEM (Patient-Oriented Eczema Measure) by the first on-treatment assessment at Week 16
- Dermatology health-related quality of life (DLQI) scores by the first on-treatment assessment at Week 8
- Patient global assessment of change at Week 16, with statistically significant improvements in patients treated with 250 mg Q4W or 250 mg Q2W of lebrikizumab, respectively, rating their atopic dermatitis as "1, much better" compared with patients treated with placebo.
"These data help us understand how the treatment may work for patients to alleviate symptoms such as itch and sleep which are known to severely impact the lives of the millions of patients living with atopic dermatitis," said Lotus Mallbris, M.D., Ph.D., vice president of immunology development at Lilly. "We are currently evaluating lebrikizumab in a broad Phase 3 clinical program and look forward to further progressing this important investigational medicine given the clear need for additional options for patients living with this debilitating condition."
"Itch is a debilitating symptom for people living with atopic dermatitis, and it affects the physical, mental, emotional, social and financial areas in their lives," said Lisa Butler, senior vice president, strategic partnerships and programs at the National Eczema Association. "The burden on patients and families is still widely underappreciated, so it is encouraging to see new therapies in development that could potentially address the symptoms that this community struggles with the most on a daily basis."
In the randomized, double-blind, placebo-controlled, Phase 2b dose-ranging study, 280 patients with moderate-to-severe atopic dermatitis were randomized 3:3:3:2 to one of three doses of subcutaneous lebrikizumab (125 mg every four weeks, 250 mg every four weeks, or 250 mg every two weeks) or placebo every two weeks for 16 weeks with safety follow-up to Week 32. The primary endpoint was the percent change in Eczema Area Severity Index (EASI) from baseline at Week 16. Secondary endpoints measured atopic dermatitis severity, itch, sleep loss, and included the proportions of patients achieving EASI50, EASI75, EASI90, a score of 0 or 1 on the Investigator's Global Assessment (IGA), and a >4-point improvement on the pruritus (itch) 11-point numeric rating scale (NRS) assessing daily itch. Change in sleep loss from baseline also was measured.
Lebrikizumab was generally well-tolerated. The safety profile was consistent with previous studies, including low frequency of conjunctivitis, herpes virus infections and injection site reactions.
About Atopic Dermatitis
Atopic dermatitis is the most common and severe form of eczema, a chronic inflammatory condition that can present as early as childhood and continue into adulthood. A moderate-to-severe form of the disease is characterized by a range of signs and symptoms, including rashes on the skin that often cover much of the body, as well as intense, persistent itching. The condition can have a negative impact on patients' mental and physical functioning, limiting their daily activities and health-related quality of life. Patients with moderate-to-severe atopic dermatitis have reported a larger impact on quality of life than patients with psoriasis.
About Lebrikizumab
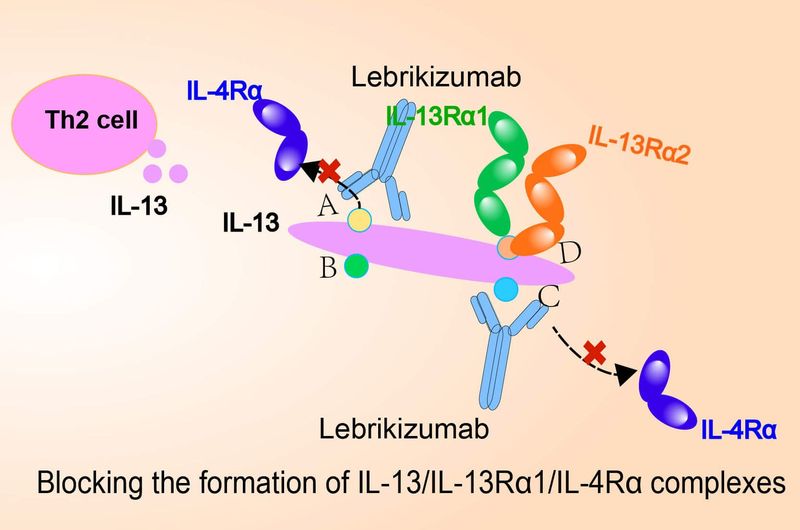
Lebrikizumab is a novel, investigational, monoclonal antibody designed to bind IL-13 with very high affinity to specifically prevent the formation of the IL-13Rα1/IL-4Rα heterodimer complex and subsequent signaling, thereby inhibiting the biological effects of IL-13 in a targeted and efficient fashion. IL-13 is believed to be a central pathogenic mediator that drives multiple aspects of the pathophysiology underlying the range of signs and symptoms of atopic dermatitis by promoting type 2 inflammation and mediating its effects on tissue, resulting in skin barrier dysfunction, itch, skin thickening and infection.
Lebrikizumab is currently being evaluated in five Phase 3 studies. The ADvocate 1 and ADvocate 2 monotherapy studies and ADhere study in combination with topical corticosteriods are to confirm its safety and efficacy in adolescent and adult patients, ages 12 years and older and 40 kg or greater, with moderate-to-severe atopic dermatitis, along with the ADore adolescent open label safety study and ADjoin long term extension study.
The U.S. Food and Drug Administration (FDA) has granted Fast Track designation to lebrikizumab for moderate-to-severe atopic dermatitis in patients aged 12 years and older and 40 kg or greater.
About Lilly in Dermatology
By following the science through unchartered territory, we continue Lilly's legacy of delivering innovative medicines that address unmet needs and have significant impacts on people's lives around the world. Skin-related diseases are more than skin deep. We understand the devastating impact this can have on people's lives. At Lilly, we are relentlessly pursuing a robust dermatology pipeline to provide innovative, patient-centered solutions so patients with skin-related diseases can aspire to live life without limitations.
About Eli Lilly and Company
Lilly is a global healthcare leader that unites caring with discovery to create medicines that make life better for people around the world. We were founded more than a century ago by a man committed to creating high-quality medicines that meet real needs, and today we remain true to that mission in all our work. Across the globe, Lilly employees work to discover and bring life-changing medicines to those who need them, improve the understanding and management of disease, and give back to communities through philanthropy and volunteerism. To learn more about Lilly, please visit us at www.lilly.com and newsroom.lilly.com/social-channels. P-LLY
About Dermira
Dermira, Inc., a wholly-owned subsidiary of Eli Lilly and Company, is a biopharmaceutical company dedicated to bringing biotech ingenuity to medical dermatology by delivering differentiated, new therapies to the millions of patients living with chronic skin conditions. Dermira is committed to understanding the needs of both patients and physicians and using its insight to identify, develop and commercialize leading-edge medical dermatology products. The company's approved treatment, QBREXZA® (glycopyrronium) cloth, is indicated for pediatric and adult patients (ages 9 and older) with primary axillary hyperhidrosis (excessive underarm sweating). Please see the QBREXZA prescribing information. Dermira is headquartered in Menlo Park, Calif. For more information, please visit http://www.dermira.com. Follow Dermira on Twitter, LinkedIn and Instagram.
SOURCE Eli Lilly and Company
Posted: June 2020

