Apellis Announces Plans to Submit NDA for Pegcetacoplan in Paroxysmal Nocturnal Hemoglobinuria (PNH)
WALTHAM, Mass., May 21, 2020 (GLOBE NEWSWIRE) -- Apellis Pharmaceuticals, Inc. (Nasdaq: APLS), a global biopharmaceutical company pioneering targeted C3 therapies, today announced plans to submit a New Drug Application (NDA) for pegcetacoplan for the treatment of paroxysmal nocturnal hemoglobinuria (PNH) in the second half of 2020. Based on feedback from the U.S. Food and Drug Administration (FDA) during a pre-NDA meeting to discuss the registration strategy for pegcetacoplan, Apellis plans to submit the NDA with results from the Phase 3 PEGASUS trial comparing pegcetacoplan to eculizumab in patients with PNH.
“We are pleased with the successful outcome of the pre-NDA meeting and committed to working closely with the FDA and other regulatory authorities to bring pegcetacoplan to the PNH community,” said Federico Grossi, M.D., Ph.D., Chief Medical Officer of Apellis. “Based on results from our Phase 3 PEGASUS study, we are excited about the potential for pegcetacoplan to redefine treatment for people living with PNH.”
Regulatory discussions are also ongoing in the EU, and Apellis plans to meet with European regulatory agencies in the second quarter of 2020 to discuss next steps for submission of a marketing authorization application for pegcetacoplan in PNH.
The PEGASUS study met its primary endpoint, demonstrating the superiority of pegcetacoplan to eculizumab with a statistically significant improvement in hemoglobin levels at 16 weeks. Top-line results also showed that the safety profile of pegcetacoplan was comparable to eculizumab in the study. As previously announced, results from the PEGASUS study have been selected for an oral presentation at the 25th Congress of the European Hematology Association and will be available on June 12, 2020.
About the PEGASUS Study
The PEGASUS study (APL2-302; NCT03500549) is a multi-center, randomized, open-label, active-comparator controlled Phase 3 study in 80 adults with PNH. The primary objective of this study was to establish the efficacy and safety of pegcetacoplan compared to eculizumab. Participants must have been on eculizumab (stable for at least 3 months) with a hemoglobin level of <10.5 g/dL at the screening visit. During the four-week run-in, patients were dosed with 1080 mg of pegcetacoplan twice weekly in addition to their current dose of eculizumab. During the 16-week randomized, controlled period, patients were randomized to receive either 1080 mg of pegcetacoplan twice weekly or their current dose of eculizumab. All participants completing the randomized controlled period entered the open-label pegcetacoplan treatment period where they received pegcetacoplan, regardless of the prior treatment received in the randomized, controlled period.
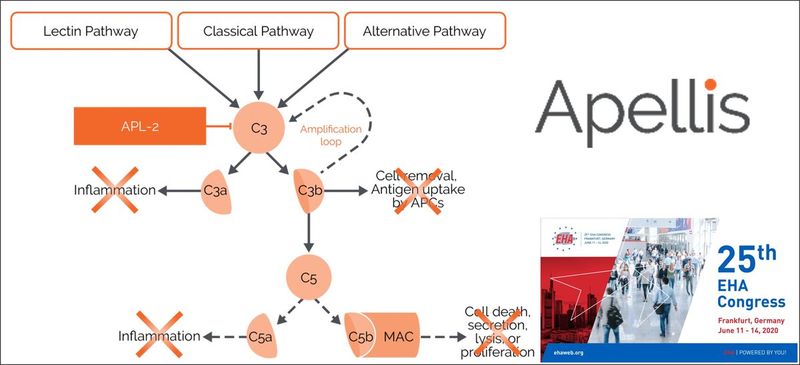
About Pegcetacoplan (APL-2)
Pegcetacoplan is an investigational, targeted C3 inhibitor designed to regulate excessive complement activation, which can lead to the onset and progression of many serious diseases. Pegcetacoplan is a synthetic cyclic peptide conjugated to a polyethylene glycol polymer that binds specifically to C3 and C3b. Apellis is evaluating pegcetacoplan in several clinical studies including paroxysmal nocturnal hemoglobinuria (PNH), geographic atrophy (GA), cold agglutinin disease, and C3 glomerulopathy. Pegcetacoplan was granted Fast Track designation by the U.S. Food and Drug Administration (FDA) for the treatment of PNH and the treatment of GA. For additional information regarding our clinical trials, visit www.apellis.com/clinical-trials.html.
About Paroxysmal Nocturnal Hemoglobinuria (PNH)
PNH is a rare, chronic, life-threatening blood disorder associated with abnormally low hemoglobin levels due to the destruction of oxygen-carrying red blood cells (hemolysis). Persistently low hemoglobin can result in frequent transfusions and debilitating symptoms such as severe fatigue and difficulty breathing (dyspnea). Retrospective studies show that, even on eculizumab, approximately 70% of people with PNH have low hemoglobin levels,1,2 and 36% require one or more transfusions a year.2
About Apellis
Apellis Pharmaceuticals, Inc. is a global biopharmaceutical company that is committed to leveraging courageous science, creativity, and compassion to deliver life-changing therapies. By pioneering targeted C3 therapies, we aim to develop best-in-class and first-in-class therapies for a broad range of debilitating diseases that are driven by uncontrolled or excessive activation of the complement cascade, including those within hematology, ophthalmology, and nephrology. For more information, please visit http://apellis.com.
Source: Apellis Pharmaceuticals, Inc.
Posted: May 2020

