Kadmon Announces Positive Topline Results of Pivotal Trial of Belumosudil (KD025) in Chronic Graft-Versus-Host Disease
NEW YORK, NY / ACCESSWIRE / May 21, 2020 / -- Kadmon Holdings, Inc. (NYSE:KDMN) today announced positive topline results from the primary analysis of ROCKstar (KD025-213), the pivotal trial evaluating belumosudil (KD025) in patients with chronic graft-versus-host disease (cGVHD) who have received at least two prior lines of systemic therapy.
Belumosudil (KD025) achieved clinically meaningful and statistically significant Overall Response Rates (ORRs) of 73% with 200 mg once daily (QD) (95% Confidence Interval (CI): 60%, 83%; p<0.0001) and 74% with 200 mg twice daily (BID) (95% CI: 62%, 84%; p<0.0001). Responses were achieved across key patient subgroups and complete responses were observed in all organ systems.
The study had previously met the primary endpoint of Overall Response Rate (ORR) at the interim analysis, which was conducted as scheduled as two months after completion of enrollment and was previously reported. Today's results from the primary analysis, which was conducted six months after completion of enrollment, show even greater efficacy of belumosudil (KD025) in cGVHD patients.
While data continue to mature, 49% of responders have maintained their response for at least 20 weeks at the time of the primary analysis. With a median treatment duration of 29 weeks, the median duration of response has not yet been reached in this ongoing study.
Belumosudil has been well tolerated and adverse events have been consistent with those expected in the patient population. No cytomegalovirus (CMV) infection or reactivation has been observed and no significant drug-related cytopenias have been reported.
Kadmon is submitting its New Drug Application (NDA) to the U.S. Food and Drug Administration (FDA) under the Real-Time Oncology Review (RTOR) pilot program, an initiative of the FDA's Oncology Center of Excellence. Kadmon is on track to complete its NDA submission for belumosudil (KD025) in the fourth quarter of 2020.
"Belumosudil has shown robust and durable responses across the spectrum of this difficult-to-treat disease and we are extraordinarily pleased with this outcome," said Harlan W. Waksal, M.D., President and CEO of Kadmon. "We are excited to participate in the FDA's Real-Time Oncology Review pilot program, which seeks to promote efficient review of much-needed treatments. We continue to work closely with the Agency through their review of our data as we prepare to complete our NDA submission."
"The remarkable results achieved with belumosudil in this advanced patient population demonstrate its broad potential to offer meaningful clinical benefit to cGVHD patients," said Sanjay K. Aggarwal, M.D., Senior Vice President, Clinical Development of Kadmon. "We look forward to sharing the full dataset from the ROCKstar study at an upcoming medical meeting."
"Treatment with belumosudil has demonstrated compelling results for this major unmet medical need. Importantly, belumosudil has been very well tolerated, which allows trial participants to stay on therapy and achieve meaningful responses," said Madan Jagasia, MD, MS, MMHC, Professor of Medicine, Vanderbilt University Medical Center; Co-Leader, Translational Research and Interventional Oncology; Chief Medical Officer, Vanderbilt-Ingram Cancer Center, an investigator of the KD025-208 and ROCKstar studies and the ROCKstar Steering Committee chair. "These results demonstrate the potential of belumosudil to become a cornerstone of the cGVHD treatment paradigm, if approved, as it delivers meaningful and sustained benefits to patients with this serious condition."
About ROCKstar
ROCKstar (KD025-213) is an ongoing open-label trial of belumosudil (KD025) in adults and adolescents with cGVHD who have received at least two prior lines of systemic therapy. Patients were randomized to receive belumosudil (KD025) at 200 mg QD or 200 mg BID, enrolling 66 patients per arm. The primary endpoint of the study is Overall Response Rate (ORR). Statistical significance is achieved if the lower bound of the 95% CI of ORR exceeds 30%. The ORR endpoint was met at the interim analysis, conducted two months after completion of enrollment, with ORRs of 64% and 67% in the respective arms. At the study's primary analysis, conducted six months after completion of enrollment, belumosudil achieved ORRs of 73% and 74% in the respective arms. Kadmon plans to submit results from the ROCKstar primary analysis for presentation at an upcoming scientific meeting.
About Belumosudil (KD025)
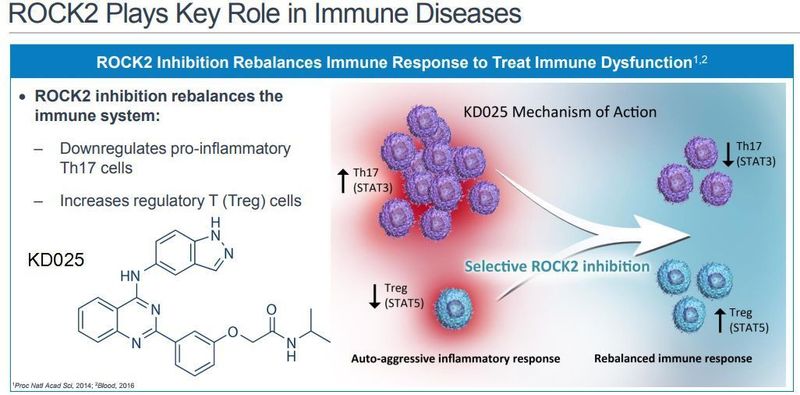
Belumosudil (KD025) is a selective oral inhibitor of Rho-associated coiled-coil kinase 2 (ROCK2), a signaling pathway that modulates inflammatory response. In an earlier Phase 2 study of belumosudil in cGVHD patients (KD025-208), belumosudil achieved an ORR of 65%. Data from this study led to FDA Breakthrough Therapy Designation by the FDA for belumosudil for the treatment of patients with cGVHD who have received at least two prior lines of systemic therapy. The FDA has also granted Orphan Drug Designation to belumosudil for the treatment of cGVHD. Kadmon is submitting its NDA for belumosudil under the FDA's Real-Time Oncology Review (RTOR) pilot program. In addition to cGVHD, belumosudil is being studied in an ongoing Phase 2 clinical trial in adults with diffuse cutaneous systemic sclerosis (KD025-209).
About cGVHD
cGVHD is a common and often fatal complication following hematopoietic stem cell transplantation. In cGVHD, transplanted immune cells (graft) attack the patient's cells (host), leading to inflammation and fibrosis in multiple tissues, including skin, mouth, eye, joints, liver, lung, esophagus and gastrointestinal tract. Approximately 14,000 patients in the United States are currently living with cGVHD in the United States.
About Kadmon
Kadmon is a biopharmaceutical company developing innovative products for significant unmet medical needs. Our product pipeline is focused on inflammatory and fibrotic diseases as well as immuno-oncology.
SOURCE: Kadmon Holdings, Inc.
Posted: May 2020

