FDA Grants Accelerated Approval for Ojemda (tovorafenib) for Relapsed or Refractory BRAF-altered Pediatric Low-Grade Glioma
BRISBANE, Calif., April 23, 2024 (GLOBE NEWSWIRE) -- Day One Biopharmaceuticals, Inc. (Nasdaq: DAWN) (“Day One” or the “Company”), a commercial-stage biopharmaceutical company dedicated to developing and commercializing targeted therapies for people of all ages with life-threatening diseases, today announced that the U.S. Food and Drug Administration (FDA) has approved Ojemda (tovorafenib), a type II RAF inhibitor, for the treatment of patients 6 months of age and older with relapsed or refractory pLGG harboring a BRAF fusion or rearrangement, or BRAF V600 mutation. This indication is approved under accelerated approval based on response rate and duration of response. With the approval, Day One received a rare pediatric disease priority review voucher from the FDA.
“Ojemda ushers in a new day for children living with relapsed or refractory pLGG, and we are pleased that we can deliver a new medicine for these patients in desperate need of new treatment options. Moreover, Ojemda is the first and only FDA-approved medicine for children with BRAF fusions or rearrangements, which are the most common molecular alteration in pLGG,” said Jeremy Bender, Ph.D., chief executive officer of Day One. “We are very proud that our first approved medicine addresses this serious and life-threatening disease of childhood and adolescence. We are grateful to the pLGG community, including patients and their families, study investigators, non-profit organizations, and advocacy groups, for their collaboration and support as we strive to close the innovation gap for children with cancer awaiting new treatments.”
pLGG is the most common brain tumor diagnosed in children, with patients suffering profound tumor- and treatment-associated morbidities that can impact their life trajectory. BRAF is the most commonly altered gene in pLGG, with up to 75 percent of children having a BRAF alteration. Until now, there had been no medicines approved for patients with pLGG driven by BRAF fusions.
“pLGG is a chronic and relentless cancer that can devastate children and their families, often stealing their vision, balance and speech,” said Dr. Sabine Mueller, pediatric neuro-oncologist, University of California San Francisco Benioff Children’s Hospitals. “The goal of pLGG treatment is to stabilize or shrink the tumor without further disrupting the child’s and family’s life. Historically, there has been no standard of care for children with pLGG who have relapsed. We are excited to welcome a new targeted treatment option with once-weekly oral dosing designed specifically for these kids and their families.”
Ojemda is the only systemic therapy for pLGG that offers once-weekly dosing, with or without food, as a tablet or oral suspension.
Approval Based on Multiple Criteria
The accelerated approval of Ojemda is based on data from the Company’s pivotal open-label Phase 2 FIREFLY-1 trial, which enrolled a total of 137 relapsed or refractory BRAF-altered pLGG patients across two study arms. Arm 1, which accrued 77 patients, was used for the efficacy analyses. Arm 2 provided additional safety data from an incremental 60 patients and was initiated to enable access to tovorafenib once Arm 1 had fully accrued. Details of this trial were presented in November 2023 at the Society for Neuro-Oncology meeting through two oral plenary presentations and in parallel through a publication in Nature Medicine.
The approval of Ojemda was based, in part, on the major efficacy outcome measure of overall response rate (ORR), defined as the proportion of patients with complete response (CR), partial response (PR), or minor response (MR) by independent review based on Response Assessment in Pediatric Neuro-Oncology Low-Grade Glioma (RAPNO LGG).
“This is a tremendous moment not only for Day One, but also for the broader pediatric brain tumor community. Thanks to the close collaboration between RAPNO and Response Assessment for Neuro-Oncology (RANO) working groups and the patients and families impacted by a pLGG diagnosis, the way we think about measuring response and the goals of therapy for this unique patient population truly evolved,” said Dr. Samuel Blackman, co-founder and head of research and development at Day One. “As a pediatric neuro-oncologist, the approval of Ojemda is a dream realized.”
In Arm 1, data from the 76 RAPNO LGG evaluable patients include:
- A best ORR of 51% (95% CI: 40 - 63), which included 28% PRs and 11% MRs.
- The ORR for Ojemda was 52% among the 64 patients with BRAF fusions or rearrangements and 50% for the 12 patients with a BRAF V600 mutation.
- The ORR was 49% among the 45 patients who had received a prior MAPK-targeted therapy, and 55% among the 31 patients who had not received a prior MAPK-targeted therapy.
- As of the June 5, 2023 data cutoff, the median duration of response by RAPNO LGG was 13.8 months (95% CI: 11.3, not estimable). In addition, 66% of patients remained on study and continue on treatment as of this date.
- The median time to response, following initiation of treatment, with Ojemda was 5.3 months (range 1.6, 11.2).
- Based on RANO LGG criteria, the ORR was 53% [95% CI: (41, 64)].
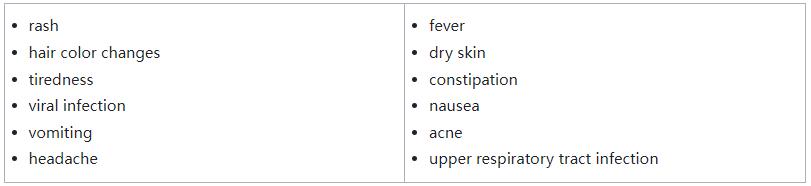
The safety of Ojemda was evaluated in 137 patients with relapsed or refractory pLGG, with the majority of adverse events being Grade 1 or Grade 2. The most common side effects were rash, hair color changes, tiredness, viral infection, vomiting, headache, fever, dry skin, constipation, nausea, acne and upper respiratory tract infection.
“This is an exciting moment for children and families living with pLGG who previously had few treatment options if their disease progressed,” said Courtney Davies, president and chief executive officer of the Pediatric Brain Tumor Foundation. “The approval of Ojemda is a testament to the power of community and industry collaboration to address a critical unmet need for children whose day-to-day living and long-term health outcomes are significantly impacted by pLGG. The potential benefit that a new treatment option provides children living with this disease and their families is crucial. There is so much to celebrate here.”
The Company continues its commitment to the pLGG community with the Phase 3 FIREFLY-2/LOGGIC randomized clinical trial evaluating tovorafenib as a potential front-line therapy compared to chemotherapy in patients aged 6 months to 25 years with pLGG, which it believes will satisfy certain post-marketing requirements to the FDA. This study is currently enrolling patients in the United States, Canada, Europe, Australia, and Asia.
Introducing EveryDay Support From Day One™
Day One Biopharmaceuticals is dedicated to helping patients with pLGG access Ojemda and supporting their families throughout the treatment journey. As part of this commitment, we are pleased to announce EveryDay Support From Day One™, a comprehensive program that offers personalized services for eligible patients and their care teams, including insurance coverage support, financial assistance options, shipping medication to patients' homes, and educational resources. Caregivers and healthcare providers can visit www.everydaysupport.com or call a patient navigator at 855-DAY1-BIO (855-329-1246) for more information.
Ojemda will be available in the U.S. through specialty pharmacy partners Biologics and Onco360.
About Ojemda™
Ojemda (tovorafenib) is a Type II RAF kinase inhibitor of mutant BRAF V600, wild-type BRAF, and wild-type CRAF kinases.
Ojemda is indicated for the treatment of patients 6 months of age and older with relapsed or refractory pediatric low-grade glioma (LGG) harboring a BRAF fusion or rearrangement, or BRAF V600 mutation. This indication is approved under accelerated approval based on response rate and duration of response. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial(s).
Tovorafenib is under evaluation as a therapy for patients with pLGG requiring front-line treatment (Phase 3 FIREFLY-2/LOGGIC). It is also being studied in combination with the MEK inhibitor pimasertib for adolescent and adult patient populations with recurrent or progressive solid tumors with MAPK pathway alterations (FIRELIGHT-1).
Tovorafenib was granted Breakthrough Therapy and Rare Pediatric Disease designations by the FDA for the treatment of patients with pLGG harboring an activating RAF alteration, and it was evaluated by the FDA under priority review. Tovorafenib has also received Orphan Drug designation from the FDA for the treatment of malignant glioma and from the European Commission for the treatment of glioma.
For more information, please visit www.Ojemda.com.
INDICATION
What is Ojemda™ (tovorafenib)?
Ojemda is a prescription medicine used to treat certain types of brain tumors (cancers) called gliomas in patients 6 months and older:
- that is a pediatric low-grade glioma (LGG), and
- that has come back after previous treatment or has not responded to previous treatment and
- that has a certain type of abnormal “BRAF” gene.
IMPORTANT SAFETY INFORMATION
Before taking or giving Ojemda, tell your healthcare provider about all of your or your child’s medical conditions, including if you:
- have bleeding, skin, or liver problems
- are pregnant or plan to become pregnant. Ojemda can harm your unborn baby.
Females who are able to become pregnant:
- You should use effective non-hormonal birth control (contraception) during treatment with Ojemda and for 28 days after your last dose of Ojemda.
Males with female partners who are able to become pregnant should use effective non-hormonal birth control (contraception) during treatment with Ojemda and for 2 weeks after your last dose of Ojemda.
- are breastfeeding or plan to breastfeed. Do not breastfeed during treatment and for 2 weeks after your last dose of Ojemda.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
What should I avoid while taking Ojemda?
Limit the amount of time you spend in sunlight. Ojemda can make your skin sensitive to the sun (photosensitivity). Use sun protection measures, such as sunscreen, sunglasses and wear protective clothes that cover your skin during your treatment with Ojemda.
What are the possible side effects of Ojemda?
Ojemda may cause serious side effects, including:
- bleeding problems (hemorrhage) are common and can also be serious. Tell your healthcare provider if you have any signs or symptoms of bleeding, including:
- headache, dizziness or feeling weak
- coughing up blood or blood clots
- vomiting blood or your vomit looks like “coffee grounds”
- red or black stools that look like tar
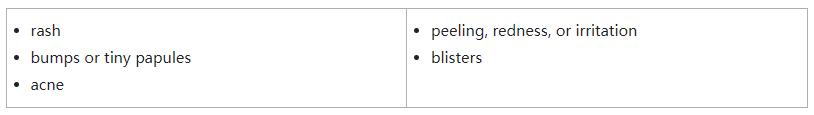
- skin reactions, including sensitivity to sunlight (photosensitivity). Ojemda can cause skin reactions that can become severe. Tell your healthcare provider if you get new or worsening skin reactions, including:
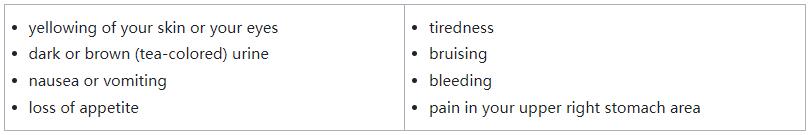
- liver problems. Your healthcare provider will do blood tests to check your liver function before and during treatment with Ojemda. Tell your healthcare provider right away if you develop any of the following symptoms:
- slowed growth in children. Growth will be checked routinely during treatment with Ojemda.
The most common side effects of Ojemda include:
Ojemda may cause fertility problems in males and females, which may affect your ability to have children.
These are not all the possible side effects of Ojemda. Call your healthcare provider for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
About Pediatric Low-Grade Glioma
Pediatric low-grade glioma (pLGG) is the most common brain tumor with an estimated incidence of 1,100 children per year who are eligible for front-line systemic therapy. BRAF is the gene most commonly altered in pLGG, of which there are two primary types of BRAF alterations – a BRAF gene fusion and a BRAF point mutation. In children with BRAF-altered pLGG, approximately 80 percent have BRAF fusions or rearrangements, while the remaining 20 percent have a V600 mutation.
Pediatric low-grade gliomas can be chronic and relentless, with patients suffering profound side effects from both the tumor and the treatment, which may include chemotherapy and radiation. These side effects can impact their life over the long term, and may include muscle weakness, loss of vision, and difficulty speaking. This type of tumor has a high risk of progression, and many children with pLGG require long-term treatment. While most children with pLGG survive their cancer, children who do not achieve a complete resection following surgery may face years of increasingly aggressive treatment.
About FIREFLY-1
FIREFLY-1 is evaluating Ojemda™ (tovorafenib) as once-weekly monotherapy in patients aged 6 months to 25 years with relapsed or progressive pLGG harboring a known activating BRAF alteration. The trial is being conducted in collaboration with the Pacific Pediatric Neuro-Oncology Consortium (PNOC). The primary endpoint is overall response rate (ORR), defined as the proportion of patients with confirmed response based upon Response Assessment for Neuro-Oncology High-Grade Glioma (RANO HGG) criteria. Secondary and exploratory endpoints include the overall response rate based on Response Assessment in Pediatric Neuro-Oncology Low-Grade Glioma (RAPNO LGG) criteria, Response Assessment for Neuro-Oncology Low-Grade Glioma (RANO LGG) criteria and volumetric analyses, progression-free survival, safety, functional outcomes, and quality of life measures. RANO HGG, RANO LGG and RAPNO LGG are assessed by blinded independent central review. Additional information about FIREFLY-1 may be found at ClinicalTrials.gov, using Identifier NCT04775485.
About the Pacific Pediatric Neuro-Oncology Consortium
The Pacific Pediatric Neuro-Oncology Consortium (PNOC) is an international consortium with study sites within the United States, Canada, Europe and Australia dedicated to bringing new therapies to children and young adults with brain tumors.
About Day One Biopharmaceuticals
Day One Biopharmaceuticals is a commercial-stage biopharmaceutical company that believes when it comes to pediatric cancer, we can do better. The Company was founded to address a critical unmet need: the dire lack of therapeutic development in pediatric cancer. Inspired by “The Day One Talk” that physicians have with patients and their families about an initial cancer diagnosis and treatment plan, Day One aims to re-envision cancer drug development and redefine what’s possible for all people living with cancer—regardless of age—starting from Day One.
Day One partners with leading clinical oncologists, families, and scientists to identify, acquire, and develop important targeted cancer treatments. The Company’s pipeline includes tovorafenib (Ojemda™) and pimasertib.
Day One is based in Brisbane, California. For more information, please visit www.dayonebio.com or find the Company on LinkedIn or X.
Cautionary Note Regarding Forward-Looking Statements
This press release contains “forward-looking” statements within the meaning of the “safe harbor” provisions of the Private Securities Litigation Reform Act of 1995, including, but not limited to: Day One’s plans to develop cancer therapies, expectations from current clinical trials, the execution of the Phase 2 and Phase 3 clinical trial for tovorafenib as designed, any expectations about safety, efficacy, timing and ability to complete clinical trials, release data results and to obtain regulatory approvals for tovorafenib and other candidates in development, and the ability of tovorafenib to treat pLGG or related indications.
Statements including words such as “believe,” “plan,” “continue,” “expect,” “will,” “develop,” “signal,” “potential,” or “ongoing” and statements in the future tense are forward-looking statements. These forward-looking statements involve risks and uncertainties, as well as assumptions, which, if they do not fully materialize or prove incorrect, could cause our results to differ materially from those expressed or implied by such forward-looking statements.
Forward-looking statements are subject to risks and uncertainties that may cause Day One’s actual activities or results to differ significantly from those expressed in any forward-looking statement, including risks and uncertainties in this press release and other risks set forth in our filings with the Securities and Exchange Commission, including Day One’s ability to develop, obtain regulatory approval for or commercialize any product candidate, Day One’s ability to protect intellectual property, the potential impact of global business or macroeconomic conditions, including as a result of inflation, rising interest rates, instability in the global banking system, geopolitical conflicts and the sufficiency of Day One’s cash, cash equivalents and investments to fund its operations. These forward-looking statements speak only as of the date hereof and Day One specifically disclaims any obligation to update these forward-looking statements or reasons why actual results might differ, whether as a result of new information, future events or otherwise, except as required by law.
Source: Day One Biopharmaceuticals, Inc.
Posted: April 2024