FDA Approves Ycanth (cantharidin) Topical Solution for the Treatment of Molluscum Contagiosum
WEST CHESTER, PA –Jul 21, 2023 (GLOBE NEWSWIRE) -- Verrica Pharmaceuticals Inc. (“Verrica”) (Nasdaq: VRCA), a dermatology therapeutics company developing medications for skin diseases requiring medical interventions, today announced U.S. Food and Drug Administration (FDA) approval of Ycanth ™ (cantharidin) topical solution for the treatment of molluscum contagiosum (molluscum) in adult and pediatric patients 2 years of age and older.
“We are proud to bring patients and caregivers the first FDA-approved treatment for molluscum, which is one of the largest unmet needs in medical dermatology,” commented Ted White, Verrica’s President and Chief Executive Officer. “Verrica is the first company to develop a proprietary applicator and GMP-formulation of cantharidin that allows a safe, effective and precise topical administration, and the first company to successfully gain FDA approval after conducting rigorous clinical trials to evaluate the safe and effective use of a cantharidin-based product for the treatment of molluscum. We wish to express our gratitude to the approximately 500 patients and their caregivers who participated in the pivotal Phase 3 trials as well as the investigators and health care professionals at more than 30 clinical sites. Our commercial team is vigorously preparing for commercial launch, and we look forward to working with healthcare providers to give patients and caregivers access to Ycanth ™ by September 2023.”
Mr. White continued: “Today’s approval of Ycanth ™ is a historic transformational moment in medical dermatology, as physicians, patients and caregivers have long sought a safe and effective FDA approved treatment for molluscum. Molluscum, which primarily affects children, is highly contagious and is commonly transmitted in households, schools, swimming pools and other extra-curricular settings. Since molluscum spreads through skin-to-skin contact and the sharing of contaminated objects with its viral lesions, a topical treatment with precise administration is essential towards preventing further transmission. Based on the results from Verrica’s clinical trials, the FDA found that Ycanth ™ is safe and effective for patients as young as two years old, providing an important treatment option for patients and caregivers struggling with this disease.”
Ycanth is for topical use only. Ycanth is not for oral, mucosal, or ophthalmic use. Local skin reactions at the application site were observed in 97% of subjects treated with Ycanth during clinical trials. Local skin reactions included vesiculation, pruritus, pain, discoloration, and erythema.
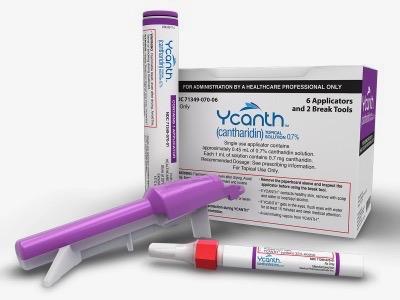
Ycanth is a proprietary drug-device combination product containing a GMP-controlled formulation of cantharidin (0.7% w/v) delivered via a single-use applicator, allowing for precise topical dosing and targeted administration. Verrica plans to make Ycanth ™ available by September 2023.
The approval is based on positive results from two identical Phase 3 randomized, double-blind, multicenter clinical trials (CAMP-1 and CAMP-2) that evaluated the safety and efficacy of VP-102 (Ycanth ™) compared to placebo in patients two years of age and older diagnosed with molluscum.
In both trials, a clinically and statistically significant number of patients treated with VP-102 met the primary endpoint of complete clearance of all treatable molluscum lesions. In CAMP-1, 46% of participants treated with VP-102 achieved complete clearance of molluscum lesions compared to 18% of participants in the vehicle group (p<0.0001); in CAMP-2, 54% of participants treated with VP-102 achieved complete clearance of molluscum lesions compared to 13% of participants in the vehicle group (p<0.0001).
Additional post-hoc analyses of the CAMP trials showed that complete clearance of all lesions was statistically significantly higher in the VP-102 group than vehicle across all body regions, including areas deemed most sensitive. An additional post-hoc analysis demonstrated that the percentage of subjects with complete molluscum clearance at the end of the trial was statistically significantly higher across all age groups for VP-102-treated subjects compared to subjects treated with vehicle.
There were no serious adverse reactions reported in the trials. Adverse reactions were mostly mild to moderate. The discontinuation rate due to an adverse reaction was 2.3% among subjects treated with Ycanth and 0.5% among subjects treated with vehicle.
About Molluscum Contagiosum (Molluscum)
Molluscum is a highly contagious viral skin disease that affects approximately six million people — primarily children — in the United States. Molluscum is caused by a pox virus that produces distinctive raised, skin-toned-to-pink-colored lesions that can cause pain, inflammation, itching and bacterial infection. It is easily transmitted through direct skin-to-skin contact or through fomites (objects that carry the disease like toys, towels or wet surfaces) and can spread to other parts of the body or to other people, including siblings. The lesions can be found on most areas of the body and may carry substantial social stigma. Without treatment, molluscum can last for an average of 13 months, and in some cases, up to several years.
About Ycanth ™
Ycanth ™ (cantharidin) topical solution is a proprietary drug-device combination product that contains a GMP-controlled formulation of cantharidin (0.7% w/v) delivered via a single-use applicator. Ycanth ™ is the first and only U.S. FDA-approved treatment for molluscum contagiosum, which is primarily a pediatric disease. The use of Ycanth ™ is supported by results from adequate and well-controlled trials in pediatric patients 2 years of age and older. The safety and efficacy in pediatric patients below the age of 2 years have not been established.
Ycanth ™ should only be administered by a trained healthcare professional. Ycanth ™ is not for home use.
Indication
Ycanth (cantharidin) topical solution, 0.7% is indicated for the topical treatment of molluscum contagiosum in adult and pediatric patients 2 years of age and older.
Important Safety Information
CONTRAINDICATIONS:
None.
WARNINGS AND PRECAUTIONS:
- Ycanth is for topical use only. Ycanth is not for oral, mucosal, or ophthalmic use. Life threatening or fatal toxicities can occur if Ycanth is administered orally. Avoid contact with the treatment area, including oral contact, after treatment. Ocular toxicity can occur if Ycanth comes in contact with eyes. If Ycanth gets in eyes, flush eyes with water for at least 15 minutes.
- Local Skin Reactions: Reactions at the application site may occur, including vesiculation, pruritus, pain, discoloration, and erythema. Avoid application near eyes and mucosal tissue, and to healthy skin. If Ycanth contacts any unintended surface, or healthy skin, immediately remove. If severe local skin reactions occur, remove prior to the recommended 24 hours after treatment.
- Ycanth is flammable, even after drying. Avoid fire, flame or smoking near lesion(s) during treatment and after application until removed.
ADVERSE REACTIONS:
The most common (incidence ≥1%) reactions are the following local skin reactions at the application site: vesiculation, pain, pruritus, scabbing, erythema, discoloration, application site dryness, edema, and erosion. Local skin reactions at the application site were observed in 97% of subjects treated with Ycanth during clinical trials. These local skin reactions are expected and related to the anticipated blistering response of the skin to cantharidin.
DRUG INTERACTIONS:
No studies evaluating the drug interaction potential of cantharidin have been conducted.
USE IN SPECIFIC POPULATIONS:
Pregnancy: There are no available data with use of Ycanth in pregnant women to evaluate for a drug-associated risk of major birth defects, miscarriage or adverse maternal or fetal outcomes. Given that systemic exposure to cantharidin following topical administration is low, maternal use is not expected to result in fetal exposure to the drug.
Lactation: Avoid application of Ycanth topical solution to areas with increased risk for potential ingestion by or ocular exposure to the breastfeeding child.
OVERDOSAGE:
Oral ingestion of cantharidin has resulted in renal failure, blistering and severe damage to the gastrointestinal tract, coagulopathy, seizures, and flaccid paralysis.
Please see accompanying full Prescribing Information.
To report SUSPECTED ADVERSE REACTIONS, contact Verrica Pharmaceuticals Inc. at 1-877-VERRICA (1-877-837-7422), or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. Local skin reactions are expected and should be reported if they are severe.
About Verrica Pharmaceuticals Inc.
Verrica is a dermatology therapeutics company developing medications for skin diseases requiring medical interventions. On July 21, 2023, Verrica’s lead product, Ycanth ™ (cantharidin) (formerly known as VP-102), became the first treatment approved by the FDA to treat pediatric and adult patients with molluscum contagiosum, a highly contagious viral skin infection affecting approximately 6 million people in the United States, primarily children. VP-102 is also in development to treat common warts and external genital warts, two of the largest unmet needs in medical dermatology. Verrica is developing VP-103, its second cantharidin-based product candidate, for the treatment of plantar warts. Verrica has also entered a worldwide license agreement with Lytix Biopharma AS to develop and commercialize VP-315 (formerly LTX-315 and VP-LTX-315) for dermatologic oncology conditions. For more information, visit www.verrica.com.
Forward-Looking Statements
Any statements contained in this press release that do not describe historical facts may constitute forward-looking statements as that term is defined in the Private Securities Litigation Reform Act of 1995. These statements may be identified by words such as “believe,” “expect,” “may,” “plan,” “potential,” “will,” and similar expressions, and are based on Verrica’s current beliefs and expectations. These forward-looking statements include expectations regarding the commercial launch of Ycanth , including the timing thereof, and the potential benefits of Ycanth and Verrica’s product candidates to patients. These statements involve risks and uncertainties that could cause actual results to differ materially from those reflected in such statements. Risks and uncertainties that may cause actual results to differ materially include uncertainties inherent in the drug development process and the regulatory approval process, Verrica’s reliance on third parties over which it may not always have full control and uncertainties that are described in Verrica’s Annual Report on Form 10-K for the year ended December 31, 2022 and other filings Verrica makes with the U.S. Securities and Exchange Commission. Any forward-looking statements speak only as of the date of this press release and are based on information available to Verrica as of the date of this release, and Verrica assumes no obligation to, and does not intend to, update any forward-looking statements, whether as a result of new information, future events or otherwise.
Source: Verrica Pharmaceuticals Inc.
Posted: July 2023