FDA Approves Columvi (glofitamab-gxbm) Bispecific Antibody for the Treatment of Relapsed or Refractory Diffuse Large B-Cell Lymphoma
South San Francisco, CA -- June 15, 2023 -- Genentech, a member of the Roche Group (SIX: RO, ROG; OTCQX: RHHBY), announced today that the U.S. Food and Drug Administration (FDA) has approved Columvi (glofitamab-gxbm) for the treatment of adult patients with relapsed or refractory (R/R) diffuse large B-cell lymphoma (DLBCL) not otherwise specified or large B-cell lymphoma (LBCL) arising from follicular lymphoma, after two or more lines of systemic therapy. This indication is approved under accelerated approval based on response rate and durability of response in the Phase I/II NP30179 study. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial. Columvi will be available in the United States in the coming weeks.
“People with diffuse large B-cell lymphoma who have gone through multiple lines of therapy have a poor prognosis and desperately need additional treatment options,” said Levi Garraway, M.D., Ph.D., chief medical officer and head of Global Product Development. “As an off-the-shelf, fixed-duration treatment providing durable response rates, we believe Columvi could change the way this aggressive lymphoma is treated, reinforcing our dedication to bringing innovative treatment options to people with critical unmet needs.”
DLBCL is an aggressive, hard-to-treat disease and is the most common form of non-Hodgkin’s lymphoma in the United States. While many people with DLBCL are responsive to treatment, the majority of those who relapse or are refractory to subsequent treatments have poor outcomes.
“Patients with relapsed or refractory diffuse large B-cell lymphoma may experience rapid progression of their cancer and often urgently need an effective treatment option that can be administered without delay,” said Krish Patel, M.D., Director of the Lymphoma Program at the Swedish Cancer Institute in Seattle and investigator of the Columvi Phase I/II NP30179 study. “Experience from clinical trials demonstrates that Columvi can provide patients with relapsed or refractory diffuse large B-cell lymphoma a chance for complete remission with a fixed-duration immunotherapy and that such remissions can potentially be sustained after the end of their treatment.”
The FDA accelerated approval is based on positive results from the Phase I/II NP30179 study of Columvi given as a fixed course for 8.5 months in 132 patients with DLBCL who had relapsed or were refractory to prior therapies, including about one-third (30%) who had received prior CAR T-cell therapy. Additionally, 83% were refractory to their most recent therapy. Results showed patients treated with fixed-duration Columvi achieved durable remission, with 56% of patients achieving an overall response (OR; 74/132 [95% confidence interval (CI): 47-65]) and 43% of patients achieving a complete response (CR; 57/132 [95% CI: 35-52]). Over two-thirds of those who responded continued to respond for at least nine months (68.5% [95% CI: 56.7-80.3]). The OR rate is the combination of CR rate (a disappearance of all signs and symptoms of cancer) and partial response rate (a decrease in the amount of cancer in the body). The median duration of response was 1.5 years (18.4 months [95% CI: 11.4-not estimable]). Data from the NP30179 study were recently published in the New England Journal of Medicine.
Among 145 patients who received Columvi in the study, the most common adverse events (AEs) were cytokine release syndrome (CRS; 70%), which may be serious or life-threatening, musculoskeletal pain (21%), fatigue (20%) and rash (20%). CRS was generally low grade (52% experienced Grade 1, and 14% experienced Grade 2).
Columvi is the first and only CD20xCD3 T-cell engaging bispecific antibody for the treatment of R/R DLBCL that is given for a defined period of time. Unlike treat-to-progression approaches where treatment is given indefinitely until the cancer progresses or the therapy cannot be tolerated, Columvi is administered in 13 intravenous infusions over a maximum of 12 cycles (including step-up dosing) or until disease progression or the treatment cannot be tolerated, whichever occurs first. After Cycle 1, Columvi is administered once every three weeks. Designed to be completed in approximately 8.5 months, Columvi offers people with R/R DLBCL a target end date for their course of treatment and the possibility of a treatment-free period. Additionally, Columvi is a chemotherapy-free treatment option that is off-the-shelf and ready for infusion. Patients are pretreated with a single dose of obinutuzumab seven days prior to starting Columvi. Patients are also given a corticosteroid, an antipyretic (fever-reducing medicine) and an antihistamine as premedications to reduce the risk of CRS.
Columvi is part of Genentech’s broad and industry-leading CD20xCD3 T-cell-engaging bispecific antibody clinical development program. This includes the Phase III STARGLO study evaluating Columvi in combination with gemcitabine and oxaliplatin (GemOx) versus rituximab in combination with GemOx in patients with DLBCL who have been treated with one or more previous therapies and are ineligible for autologous stem cell transplant. Genentech’s hematology bispecific antibody portfolio also includes Lunsumio® (mosunetuzumab-axgb), which was granted accelerated approval by the FDA in December 2022 for the treatment of adult patients with R/R follicular lymphoma after two or more lines of systemic therapy. Genentech is exploring the potential of both Columvi and Lunsumio as monotherapies and in combination with other therapies, including Polivy® (polatuzumab vedotin-piiq), in earlier lines of treatment with the goal of providing patients with long-lasting outcomes. This robust development program includes two Phase III studies: CELESTIMO, investigating Lunsumio plus lenalidomide in second line plus (2L+) FL, and SUNMO, investigating Lunsumio plus Polivy in 2L+ DLBCL. Columvi received its first worldwide approval in Canada, and the European Medicines Agency’s Committee for Medicinal Products for Human Use recently granted a positive opinion recommending its approval.
Genentech is committed to helping people access the medicines they are prescribed and will be offering comprehensive services for people prescribed Columvi to help minimize barriers to access and reimbursement. For people who qualify, Genentech plans to offer patient assistance programs through Genentech Access Solutions. More information is also available at 866-4ACCESS/866-422-2377 or http://www.Genentech-Access.com.
About the NP30179 Study
The NP30179 study [NCT03075696] is a Phase I/II, multicenter, open-label, dose-escalation and expansion study evaluating the safety, efficacy and pharmacokinetics of Columvi® (glofitamab-gxbm) in people with relapsed or refractory diffuse large B-cell lymphoma. Outcome measures include complete response rate by an independent review committee (primary endpoint), overall response rate, duration of response, progression-free survival, safety and tolerability (secondary endpoints).
About Diffuse Large B-Cell Lymphoma
Diffuse large B-cell lymphoma (DLBCL) is an aggressive (fast-growing) blood cancer and is the most common form of non-Hodgkin’s lymphoma (NHL) in the U.S. While many people with DLBCL are responsive to treatment, the majority of those who relapse or are refractory to subsequent treatments have poor outcomes. DLBCL not otherwise specified is the most common category of large B-cell lymphoma (LBCL) and accounts for about 80% or more of cases. It applies to cases that do not fall into any specific disease subgroups of LBCL.
About Large B-Cell Lymphoma Arising from Follicular Lymphoma
Follicular lymphoma (FL) is the most common slow-growing (indolent) form of non-Hodgkin’s lymphoma (NHL), accounting for about one in five cases. It typically responds well to treatment but is often characterized by periods of remission and relapse. The disease typically becomes harder to treat each time a patient relapses, and early progression can be associated with poor long-term prognosis. As part of the natural history of FL, it can transform to more aggressive malignancies, including large B-cell lymphoma (LBCL), one of the most aggressive (fast-growing) blood cancers.
About Columvi® (glofitamab-gxbm)
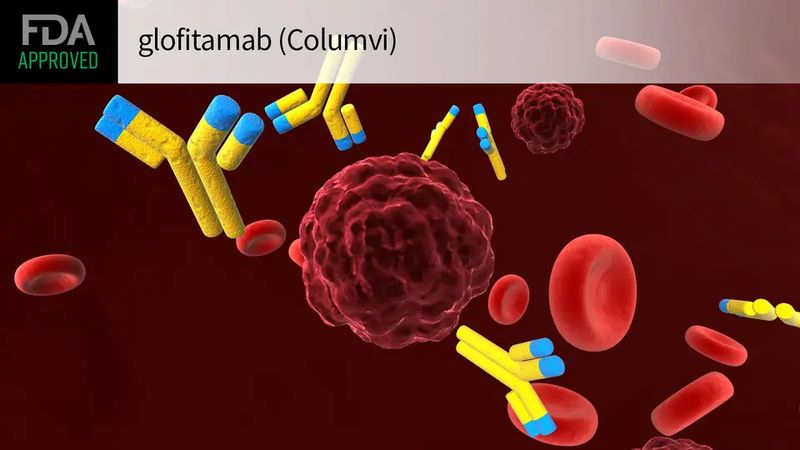
Columvi is a CD20xCD3 T-cell engaging bispecific antibody designed with a novel 2:1 structural format. Columvi targets both T cells, a type of immune cell, and B cells, which are cancerous in DLBCL. Columvi is engineered to have one region that binds to CD3, a protein on T cells, and two regions that bind to CD20, a protein on B cells, which can be healthy or cancerous. This dual-targeting brings the T cell in close proximity to the B cell, activating the release of cancer cell-killing proteins from the T cell that result in the death of B cells. A robust clinical development program for Columvi is ongoing, investigating the molecule as a monotherapy and in combination with other medicines for the treatment of people with B-cell non-Hodgkin’s lymphomas, including diffuse large B-cell lymphoma and other blood cancers.
Columvi U.S. Indication
Columvi (glofitamab-gxbm) is a prescription medicine to treat adults with certain types of diffuse large B-cell lymphoma (DLBCL) or large B-cell lymphoma (LBCL) that has come back (relapsed) or that did not respond to previous treatment (refractory), and who have received 2 or more prior treatments for their cancer.
It is not known if Columvi is safe and effective in children.
The conditional approval of Columvi is based on response rate and durability of response. There are ongoing studies to establish how well the drug works.
What is the most important information I should know about Columvi?
Columvi can cause Cytokine Release Syndrome (CRS), a serious side effect that is common during treatment with Columvi, and can also be serious and lead to death.
Call your healthcare provider or get emergency medical help right away if you develop any signs or symptoms of CRS, including:
- fever of 100.4°F (38°C) or higher
- chills or shaking
- fast or irregular heartbeat
- dizziness or light-headedness
- trouble breathing
- shortness of breath
Due to the risk of CRS, you will receive Columvi on a “step-up dosing schedule”.
- A single dose of a medicine called obinutuzumab will be given to you on the first day of your first treatment cycle (Day 1 of Cycle 1).
- You will start the Columvi step-up dosing schedule a week after the obinutuzumab dose. The step-up dosing schedule is when you receive smaller “step-up” doses of Columvi on Day 8 and Day 15 of Cycle 1. This is to help reduce your risk of CRS. You should be hospitalized during your infusion and for 24 hours after receiving the first step-up dose on Day 8. You should be hospitalized during your infusion and for 24 hours after receiving the second step-up dose on Day 15 if you experienced CRS during the first step-up dose.
- You will receive your first full dose of Columvi a week after the second step-up dose (this will be Day 1 of Cycle 2).
- If your dose of Columvi is delayed for any reason, you may need to repeat the “step-up dosing schedule”.
- If you had more than mild CRS with your previous dose of Columvi, you should be hospitalized during and for 24 hours after receiving your next dose of Columvi.
- Before each dose of Columvi, you will receive medicines to help reduce your risk of CRS and infusion-related reactions.
Your healthcare provider will monitor you for CRS during treatment with Columvi and may treat you in a hospital if you develop signs and symptoms of CRS. Your healthcare provider may temporarily stop or completely stop your treatment with Columvi if you have severe side effects.
Carry the Columvi Patient Wallet Card with you at all times and show it to all of your healthcare providers. The Columvi Patient Wallet Card lists the signs and symptoms of CRS you should get emergency medical help for right away.
What are the possible side effects of Columvi?
Columvi may cause serious side effects, including:
- Cytokine Release Syndrome.
- Neurologic problems. Columvi can cause serious neurologic problems that may lead to death. Your healthcare provider will monitor you for neurologic problems during treatment with Columvi. Your healthcare provider may also refer you to a healthcare provider who specializes in neurologic problems. Tell your healthcare provider right away if you develop any signs or symptoms of neurologic problems, including:
- headache
- confusion and disorientation
- difficulty paying attention or understanding things
- trouble speaking
- sleepiness
- memory problems
- numbness, tingling, or weakness of the hands or feet
- dizziness
- shaking (tremors)
- Serious Infections. Columvi can cause serious infections that may lead to death. Your healthcare provider will monitor you for signs and symptoms of infection and treat you as needed. Tell your health care provider right away if you develop any signs of an infection, including: fever, chills, weakness, cough, shortness of breath, or sore throat.
- Growth in your tumor or worsening of tumor related problems (tumor flare). Tell your healthcare provider if you get any of these signs or symptoms of tumor flare:
- tender or swollen lymph nodes
- pain or swelling at the site of the tumor
- chest pain
- cough
- trouble breathing
The most common side effects of Columvi include: CRS, muscle and bone pain, rash, and tiredness.
The most common severe abnormal lab test results with Columvi include: decreased white blood cells, decreased phosphate (an electrolyte), increased uric acid levels, and decreased fibrinogen (a protein that helps with blood clotting).
Your healthcare provider may temporarily stop or completely stop treatment with Columvi if you develop certain side effects.
Before receiving Columvi, tell your healthcare provider about all of your medical conditions, including if you:
- have an infection
- have kidney problems
- are pregnant or plan to become pregnant. Columvi may harm your unborn baby
Females who are able to become pregnant:
- Your healthcare provider should do a pregnancy test before you start treatment with Columvi.
- You should use effective birth control (contraception) during treatment and for 1 month after your last dose of Columvi. Talk to your healthcare provider about what birth control method is right for you during this time.
- Tell your healthcare provider right away if you become pregnant or think you may be pregnant during treatment with Columvi.
- are breastfeeding or plan to breastfeed. Columvi may pass into your breast milk. Do not breastfeed during treatment and for 1 month after your last dose of Columvi.
Tell your health care provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
What should I avoid while receiving Columvi?
Do not drive, operate heavy machinery, or do other dangerous activities if you develop dizziness, confusion, shaking (tremors), sleepiness, or any other symptoms that impair consciousness until your signs and symptoms go away. These may be signs and symptoms of neurologic problems.
These are not all the possible side effects of Columvi. Talk to your health care provider for more information about the benefits and risks of Columvi.
You may report side effects to the FDA at (800) FDA-1088 or http://www.fda.gov/medwatch. You may also report side effects to Genentech at (888) 835-2555.
Please see Important Safety Information, including Serious Side Effects, as well as the Columvi full Prescribing Information and Medication Guide or visit https://www.Columvi.com.
About Lunsumio® (mosunetuzumab-axgb)
Lunsumio is a first-in-class CD20xCD3 T-cell engaging bispecific antibody designed to target CD20 on the surface of B cells and CD3 on the surface of T cells. This dual targeting activates and redirects a patient’s existing T cells to engage and eliminate target B cells by releasing cytotoxic proteins into the B cells. A robust clinical development program for Lunsumio is ongoing, investigating the molecule as a monotherapy and in combination with other medicines, for the treatment of people with B-cell non-Hodgkin’s lymphomas, including follicular lymphoma and diffuse large B-cell lymphoma, and other blood cancers.
Lunsumio U.S. Indication
Lunsumio (mosunetuzumab-axgb) is a prescription medicine used to treat adults with follicular lymphoma whose cancer has come back or did not respond to previous treatment, and who have already received two or more treatments for their cancer.
It is not known if Lunsumio is safe and effective in children.
The conditional approval of Lunsumio is based on response rate. There are ongoing studies to establish how well the drug works.
What is the most important information I should know about Lunsumio?
Lunsumio may cause Cytokine Release Syndrome (CRS), a serious side effect that is common during treatment with Lunsumio and can also be severe or life-threatening.
Get medical help right away if you develop any signs or symptoms of CRS at any time, including:
- fever of 100.4°F (38°C) or higher
- chills
- low blood pressure
- fast or irregular heartbeat
- tiredness or weakness
- difficulty breathing
- headache
- confusion
- feeling anxious
- dizziness or light-headedness
- nausea
- vomiting
Due to the risk of CRS, you will receive Lunsumio on a “step-up dosing schedule.”
- The step-up dosing schedule is when you receive smaller “step-up” doses of Lunsumio on Day 1 and Day 8 of your first cycle of treatment
- You will receive a higher dose of Lunsumio on Day 15 of your first cycle of treatment
- If your dose of Lunsumio is delayed for any reason, you may need to repeat the step-up dosing schedule
- Before each dose in Cycle 1 and Cycle 2, you will receive medicines to help reduce your risk of CRS
Your healthcare provider will check you for CRS during treatment with Lunsumio and may treat you in a hospital if you develop signs and symptoms of CRS. Your healthcare provider may temporarily stop or completely stop your treatment with Lunsumio, if you have severe side effects.
What are the possible side effects of Lunsumio?
Lunsumio may cause serious side effects, including:
- Neurologic problems. Your healthcare provider will check you for neurologic problems during treatment with Lunsumio. Your healthcare provider may also refer you to a healthcare provider who specializes in neurologic problems. Tell your healthcare provider right away if you develop any signs or symptoms of neurologic problems during or after treatment with Lunsumio, including:
- headache
- numbness and tingling of the arms, legs, hands, or feet
- dizziness
- confusion and disorientation
- difficulty paying attention or understanding things
- forgetting things or forgetting who or where you are
- trouble speaking, reading, or writing
- sleepiness or trouble sleeping
- tremors
- loss of consciousness
- seizures
- muscle problems or muscle weakness
- loss of balance or trouble walking
- Serious infections. Lunsumio can cause serious infections that may lead to death. Your healthcare provider will check you for signs and symptoms of infection before and during treatment. Tell your healthcare provider right away if you develop any signs or symptoms of infection during treatment with Lunsumio, including:
- fever of 100.4° F (38° C) or higher
- chest pain
- tiredness
- shortness of breath
- painful rash
- sore throat
- pain during urination
- feeling weak or generally unwell
- Low blood cell counts. Low blood cell counts are common during treatment with Lunsumio and can also be severe. Your healthcare provider will check your blood cell counts during treatment with Lunsumio. Lunsumio may cause the following low blood cell counts:
- low white blood cell counts (neutropenia). Low white blood cells can increase your risk for infection
- low red blood cell counts (anemia). Low red blood cells can cause tiredness and shortness of breath
- low platelet counts (thrombocytopenia). Low platelet counts can cause bruising or bleeding problems
- Growth in your tumor or worsening of tumor related problems (Tumor flare). Lunsumio may cause serious or severe worsening of your tumor. Tell your healthcare provider if you develop any of these signs or symptoms of tumor flare during your treatment with Lunsumio: tender or swollen lymph nodes, chest pain, cough, trouble breathing, and pain or swelling at the site of the tumor
Your healthcare provider may temporarily stop or permanently stop treatment with Lunsumio if you develop severe side effects.
The most common side effects of Lunsumio include: tiredness, rash, fever, and headache.
The most common severe abnormal lab test results with Lunsumio include: decreased phosphate, increased glucose, and increased uric acid levels.
Before receiving Lunsumio, tell your healthcare provider about all of your medical conditions, including if you:
- have ever had an infusion reaction after receiving Lunsumio
- have an infection, or have had an infection in the past which lasted a long time or keeps coming back
- have or have had Epstein-Barr Virus
- are pregnant or plan to become pregnant. Lunsumio may harm your unborn baby. Tell your healthcare provider right away if you become pregnant or think you may be pregnant during treatment with Lunsumio
Females who are able to become pregnant:
- your healthcare provider should do a pregnancy test before you start treatment with Lunsumio
- you should use an effective method of birth control during your treatment and for 3 months after the last dose of Lunsumio
- are breastfeeding or plan to breastfeed. It is not known if Lunsumio passes into your breast milk. Do not breastfeed during treatment and for 3 months after the last dose of Lunsumio
Tell your health care provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
What should I avoid while receiving Lunsumio?
Do not drive, operate heavy machinery, or do other dangerous activities if you develop dizziness, confusion, tremors, sleepiness, or any other symptoms that impair consciousness until your signs and symptoms go away. These may be signs and symptoms of CRS or neurologic problems.
These are not all the possible side effects of Lunsumio. Talk to your health care provider for more information about the benefits and risks of Lunsumio.
You may report side effects to the FDA at (800) FDA-1088 or http://www.fda.gov/medwatch. You may also report side effects to Genentech at (888) 835-2555.
Please see Important Safety Information, including Serious Side Effects, as well as the Lunsumio full Prescribing Information and Medication Guide or visit https://www.Lunsumio.com.
About Polivy® (polatuzumab vedotin-piiq)
Polivy is a first-in-class anti-CD79b antibody-drug conjugate (ADC). The CD79b protein is expressed specifically in the majority of B cells, an immune cell impacted in some types of non-Hodgkin’s lymphoma (NHL), making it a promising target for the development of new therapies. Polivy binds to cancer cells such as CD79b and destroys these B cells through the delivery of an anti-cancer agent, which is thought to minimize the effects on normal cells. Polivy is being developed by Genentech using Seagen ADC technology and is currently being investigated for the treatment of several types of NHL.
Polivy U.S. Indication
Polivy is a prescription medicine used with other medicines (a rituximab product, cyclophosphamide, doxorubicin, and prednisone) as a first treatment for adults who have moderate to high risk diffuse large B-cell lymphoma (DLBCL), not otherwise specified (NOS) or high-grade B-cell lymphoma (HGBL).
Polivy is a prescription medicine used with other medicines, bendamustine and a rituximab product, to treat DLBCL in adults who have progressed after at least 2 prior therapies.
Important Safety Information
Possible serious side effects
Everyone reacts differently to Polivy therapy, so it’s important to know what the side effects are. Some people who have been treated with Polivy have experienced serious to fatal side effects. Your doctor may stop or adjust your treatment if any serious side effects occur. Be sure to contact your healthcare team if there are any signs of these side effects.
- Nerve problems in your arms and legs: This may happen as early as after your first dose and may worsen with every dose. Your doctor will monitor for signs and symptoms, such as changes in your sense of touch, numbness or tingling in your hands or feet, nerve pain, burning sensation, any muscle weakness, or changes to your walking pattern
- Infusion-related reactions: You may experience fever, chills, rash, breathing problems, low blood pressure, or hives within 24 hours of your infusion
- Low Blood Cell Counts: Treatment with Polivy can cause severe low blood cell counts. Your doctor will monitor your blood counts throughout treatment with Polivy
- Infections: If you have a fever of 100.4°F (38°C) or higher, chills, cough, or pain during urination, contact your healthcare team. Your doctor may also give you medication before giving you Polivy, which may prevent some infections, and will monitor your blood counts throughout treatment with Polivy. Treatment with Polivy can cause severe low blood cell counts
- Rare and serious brain infections: Your doctor will monitor closely for signs and symptoms of these types of infections. Contact your doctor if you experience confusion, dizziness or loss of balance, trouble talking or walking, or vision changes
- Tumor lysis syndrome: Caused by the fast breakdown of cancer cells. Signs include nausea, vomiting, diarrhea, and lack of energy
- Potential harm to liver: Some signs include tiredness, weight loss, pain in the abdomen, dark urine, and yellowing of your skin or the white part of your eyes. You may be at higher risk if you already had liver problems or you are taking other medication
Side effects seen most often
The most common side effects of Polivy when used as a first treatment in DLBCL with the medicines rituximab product, cyclophosphamide, doxorubicin, and prednisone include
- Nerve problems in arms and legs
- Nausea
- Tiredness or lack of energy
- Diarrhea
- Constipation
- Hair Loss
- Redness and sores of the lining of the mouth, lips, throat, and digestive tract
Polivy may lower your red or white blood cell counts and increase uric acid levels.
The most common side effects of Polivy when used in DLBCL after at least 2 prior therapies with other medicines, bendamustine and a rituximab product include
- Low blood cell counts (platelets, red blood cells, white blood cells)
- Nerve problems in arms and legs
- Tiredness or lack of energy
- Diarrhea
- Nausea
- Fever
- Decreased appetite
- Infections
Polivy may not be for everyone. Talk to your doctor if you are
- Pregnant or think you are pregnant: Data have shown that Polivy may harm your unborn baby
- Planning to become pregnant: Women should avoid getting pregnant while taking Polivy. Women should use effective contraception during treatment and for 3 months after their last Polivy treatment. Men taking Polivy should use effective contraception during treatment and for 5 months after their last Polivy treatment
- Breastfeeding: Women should not breastfeed while taking Polivy and for 2 months after the last dose
These may not be all the side effects. Talk to your healthcare provider for more information about the benefits and risks of Polivy treatment.
You may report side effects to the FDA at (800) FDA-1088 or http://www.fda.gov/medwatch. You may also report side effects to Genentech at (888) 835-2555.
Please see the full Prescribing Information and visit https://www.Polivy.com for additional Important Safety Information.
About Genentech in Hematology
For more than 20 years, Genentech has been developing medicines with the goal to redefine treatment in hematology. Today, we’re investing more than ever in our effort to bring innovative treatment options to people with diseases of the blood. For more information visit http://www.gene.com/hematology.
About Genentech
Founded more than 40 years ago, Genentech is a leading biotechnology company that discovers, develops, manufactures and commercializes medicines to treat patients with serious and life-threatening medical conditions. The company, a member of the Roche Group, has headquarters in South San Francisco, California. For additional information about the company, please visit http://www.gene.com.
Source: Genentech
Posted: June 2023