FDA Approves Rethymic (allogeneic processed thymus tissue-agdc) One-Time Regenerative Tissue-Based Therapy for Pediatric Congenital Athymia
- Rethymic is the first and only FDA-approved treatment indicated for immune reconstitution in pediatric patients with congenital athymia
- Children with congenital athymia are born without a thymus causing severe immunodeficiency and immune dysregulation – with only supportive care they typically die by age two or three
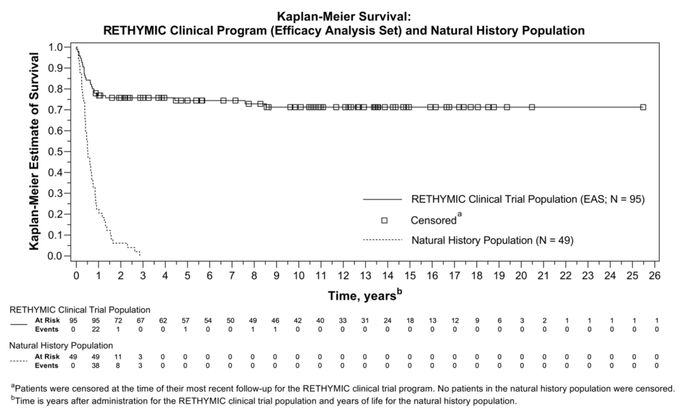
- Rethymic clinical trials included 105 patients, a data set encompassing 797 patient years of data and long-term survival to date up to 25.5 years
CAMBRIDGE, Mass. & BASEL, Switzerland, October 8, 2021 (GLOBE NEWSWIRE) – Enzyvant today announced the U.S. Food and Drug Administration (FDA) approval of Rethymic (allogeneic processed thymus tissue-agdc), a one-time regenerative tissue-based therapy for immune reconstitution in pediatric patients with congenital athymia.
“For too long, families have faced a reality that the brutal journey for pediatric congenital athymia patients receiving supportive care only would end tragically. The FDA approval of Rethymic will help patients access this desperately needed therapy beyond clinical study,” said Rachelle Jacques, CEO of Enzyvant. “We are deeply grateful to the 105 patients who participated in clinical trials, their families, and all of the stakeholders who contributed to this pioneering regenerative medicine research program.”
Pediatric congenital athymia is ultra-rare with an estimated incidence of about 17 to 24 live births each year in the United States. Children who have this condition are born without a thymus and therefore have profound immunodeficiency, life-threatening immune dysregulation, and high susceptibility to potentially fatal infections. With only supportive care, children with congenital athymia typically die by age two or three.
“This therapy is the result of more than 25 years of research aimed at increasing survival for patients who previously had very little hope,” said Louise Markert, M.D, Ph.D., principal investigator for Rethymic clinical trials and Professor of Pediatrics and Immunology at the Duke University School of Medicine. “Our research program was inspired each and every day by the possibilities that exist for children who have congenital athymia with an FDA-approved treatment for this devastating condition.”
With this FDA approval, Enzyvant has obtained a Priority Review Voucher (PRV) under the Rare Pediatric Disease Program.
Rethymic Clinical Trial Data
Ten prospective single-arm, open-label studies with patient enrollment from 1993 to 2020 form the basis of the Rethymic data set. One hundred and five patients were surgically implanted with Rethymic under one of 10 Institutional Review Board (IRB)-approved protocols. Ninety-five patients were included in the Efficacy Analysis Set (EAS) and 105 patients were included in the Safety Analysis Set.
Survival rates were analyzed with the longest follow-up period of 25.5 years. In the EAS, Kaplan-Meier estimated survival rates (95% CI) were 77% (0.670–0.841) at one year and 76% (0.658–0.832) at two years. For patients who were alive at one year post implantation, the Kaplan-Meier estimated long-term survival rate was 94% at a median follow-up time of 10.7 years. For the patients in the clinical trials, naïve T-cell levels were measured using flow cytometry at six, 12, and 24 months after implantation with Rethymic. Patients in the clinical trials started out with very few naïve T cells but naïve CD4+ and CD8+ T cells began to reconstitute over the first year, with a durable increase through year two. Reductions in the number of infections over time during the first two years after treatment were statistically significant (p<0.001).
About the Thymus and Congenital Athymia
The “T” in T cell stands for thymus because it is where T cells are selected to fight infections or are destroyed if they have the potential to attack the body instead of invaders. Congenital athymia is an ultra-rare condition in which children are born without a thymus, causing profound immunodeficiency, vulnerability to potentially fatal infections, and life-threatening immune dysregulation. With only supportive care, children with congenital athymia typically die by age two or three. Congenital athymia is initially detected by T-cell deficiency observed in newborn screening for SCID (severe combined immune deficiency), which is now required in all 50 U.S. states. SCID and congenital athymia are both primary immunodeficiency disorders but they are distinct conditions. The estimated incidence of pediatric congenital athymia in the United States is 17 to 24 live births each year.
About Rethymic
Rethymic (allogeneic processed thymus tissue-agdc) is a novel one-time tissue-based regenerative therapy used for immune reconstitution in pediatric patients with congenital athymia. Rethymic is engineered human thymus tissue designed to regenerate the thymic function children with congenital athymia are missing and does not require donor-recipient matching. Rethymic has been studied across 10 clinical trials for more than 25 years and was granted multiple U.S. Food and Drug Administration (FDA) designations including Regenerative Medicine Advanced Therapy (RMAT), Breakthrough Therapy, Rare Pediatric Disease, and Orphan Drug. It also has been granted the Orphan Drug designation and the Advanced Therapy Medicinal Product (ATMP) designation by the European Medicines Agency (EMA). Rethymic is the first and only treatment approved by the FDA for immune reconstitution in pediatric patients with congenital athymia.
Important Safety Information (ISI)
Indication
Rethymic (allogeneic processed thymus tissue–agdc) is indicated for immune reconstitution in pediatric patients with congenital athymia.
Limitations of Use:
Rethymic is not indicated for the treatment of patients with severe combined immunodeficiency (SCID).
Important Safety Information
Immune reconstitution sufficient to protect from infection is unlikely to develop prior to 6-12 months after treatment with Rethymic. Given the immunocompromised condition of athymic patients, follow infection control measures until the development of thymic function is established as measured through flow cytometry. Monitor patients closely for signs of infection including fever. If a fever develops, assess the patient by blood and other cultures and treat with antimicrobials as clinically indicated. Patients should be maintained on immunoglobulin replacement therapy until specified criteria are met, and two months after stopping, IgG trough level should be checked. Prior to and after treatment with Rethymic, patients should be maintained on Pneumocystis jiroveci pneumonia prophylaxis until specified criteria are met.
Rethymic may cause or exacerbate pre-existing graft versus host disease (GVHD). Monitor and treat patients at risk for the development of GVHD. Risk factors for GVHD include atypical complete DiGeorge anomaly phenotype, prior HCT and maternal engraftment. GVHD may manifest as fever, rash, lymphadenopathy, elevated bilirubin and liver enzymes, enteritis, and/or diarrhea.
Treatment with Rethymic may increase the risk of autoimmune-mediated conditions. These events included: thrombocytopenia, neutropenia, proteinuria, hemolytic anemia, alopecia, hypothyroidism, autoimmune hepatitis, autoimmune arthritis, transverse myelitis, albinism, hyperthyroidism, and ovarian failure. Monitor for the development of autoimmune disorders, including complete blood counts with differential, liver enzymes, serum creatinine, urinalysis, and thyroid function.
Pre-existing renal impairment is a risk factor for death.
In the clinical studies of Rethymic, 3 out of 4 patients with pre-existing cytomegalovirus infection died. The benefits/risks of treatment should be considered prior to treating patients with pre-existing CMV infection.
Because of the underlying immune deficiency, patients who receive Rethymic may be at risk of developing post-treatment lymphoproliferative disorder. Patients should be monitored for the development of lymphoproliferative disorder.
Transmission of infectious disease may occur because Rethymic is derived from human tissue and because product manufacturing includes porcine- and bovine-derived reagents.
Immunizations should not be administered in patients who have received Rethymic until immune-function criteria have been met.
All patients should be screened for anti-HLA antibodies prior to receiving Rethymic. Patients testing positive for anti-HLA antibodies should receive Rethymic from a donor who does not express those HLA alleles. HLA matching is required in patients who have received a prior hematopoietic cell transplantation (HCT) or a solid organ transplant. Patients who have received a prior HCT are at increased risk of developing GVHD after Rethymic if the HCT donor did not fully match the recipient.
Of the 105 patients in clinical studies, 29 patients died, including 23 deaths in the first year (< 365 days) after implantation.
The most common (>10%) adverse events related to Rethymic included: hypertension, cytokine release syndrome, rash, hypomagnesemia renal impairment / failure thrombocytopenia, and graft versus host disease.
To report suspected adverse reactions, please contact the FDA at 1-800-FDA-1088 or http://www.fda.gov/safety/medwatch.
About Enzyvant
Enzyvant, a wholly-owned subsidiary of Sumitovant Biopharma Ltd. (wholly-owned by Sumitomo Dainippon Pharma Co., Ltd.), is a biotechnology company dedicated to developing novel, transformative regenerative therapies for people with devastating rare diseases. Enzyvant’s Rethymic® (previously RVT-802) is a one-time tissue-based regenerative therapy approved by the U.S. Food and Drug Administration for pediatric congenital athymia, an ultra-rare and life-threatening pediatric immunodeficiency. For more information about Enzyvant, visit Enzyvant.com. Follow @Enzyvant on Twitter, Facebook, and LinkedIn.
About Sumitovant Biopharma Ltd.
Sumitovant is a global biopharmaceutical company leveraging data-driven insights to rapidly accelerate development of new potential therapies for unmet patient conditions. Through our unique portfolio of wholly-owned “Vant” subsidiaries—Urovant, Enzyvant, Spirovant, Altavant—and use of embedded computational technology platforms to generate business and scientific insights, Sumitovant has supported development of FDA-approved products including GEMTESA® for overactive bladder and Rethymic® for pediatric congenital athymia, and has advanced a promising pipeline of early-through late-stage investigational assets for other serious conditions. Sumitovant is the majority-shareholder of Myovant (NYSE: MYOV) whose marketed products include ORGOVYX® for advanced prostate cancer and MYFEMBREE® for uterine fibroids. Sumitovant is a wholly-owned subsidiary of Sumitomo Dainippon Pharma. For more information, please visit https://www.sumitovant.com.
About Sumitomo Dainippon Pharma Co., Ltd.
Sumitomo Dainippon Pharma is among the top-ten listed pharmaceutical companies in Japan, operating globally in major pharmaceutical markets, including Japan, the U.S., China, and other Asian countries. Sumitomo Dainippon Pharma is based on the merger in 2005 between Dainippon Pharmaceutical Co., Ltd., and Sumitomo Pharmaceuticals Co., Ltd. Today, Sumitomo Dainippon Pharma has more than 7,000 employees worldwide. Additional information about Sumitomo Dainippon Pharma is available through its corporate website at https://www.ds-pharma.com/.
Source: Enzyvant
Posted: October 2021