Tirzepatide Achieved Superior A1C and Body Weight Reductions Across All Three Doses Compared to Injectable Semaglutide in Adults with Type 2 Diabetes
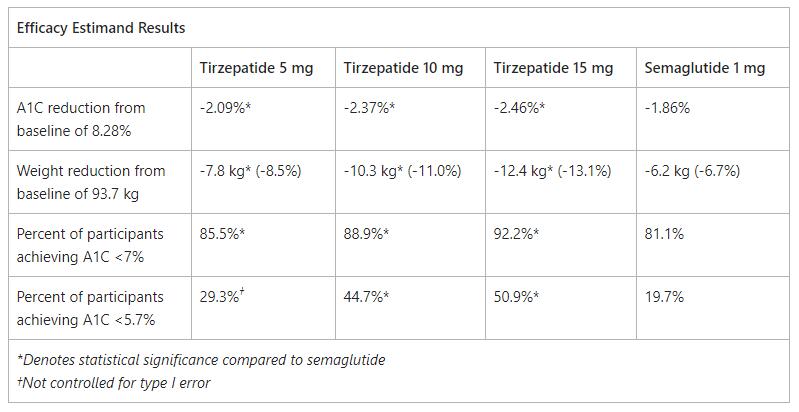
INDIANAPOLIS, March 4, 2021 /PRNewswire/ -- Tirzepatide led to superior A1C and body weight reductions from baseline across all three doses compared to injectable semaglutide 1 mg in adults with type 2 diabetes in Eli Lilly and Company's (NYSE: LLY) 40-week SURPASS-2 clinical trial. In topline results from the largest SURPASS trial to date, using the efficacy estimandi, the highest dose of tirzepatide (15 mg) reduced A1C by 2.46 percent and body weight by 12.4 kg (27.3 lb., 13.1 percent). The lowest dose of tirzepatide (5 mg) reduced A1C by 2.09 percent and body weight by 7.8 kg (17.2 lb., 8.5 percent) compared to semaglutide at 1.86 percent and 6.2 kg (13.7 lb., 6.7 percent).
Additionally, 51 percent of those taking tirzepatide 15 mg achieved an A1C of less than 5.7 percent – the level seen in people without diabetes – compared to 20 percent of those taking semaglutide. The overall safety profile of tirzepatide was similar to previously reported SURPASS trials and the well-established glucagon-like peptide-1 (GLP-1) receptor agonist class. Across all treatment arms, the most commonly reported adverse events were gastrointestinal-related.
Tirzepatide is a novel investigational once-weekly dual glucose-dependent insulinotropic polypeptide (GIP) and GLP-1 receptor agonist that integrates the actions of both incretins into a single molecule, representing a new class of medicines being studied for the treatment of type 2 diabetes. Semaglutide is a GLP-1 receptor agonist for the treatment of type 2 diabetes.
"Head-to-head studies are the gold standard for clinicians assessing the efficacy of investigational treatments, and these results show that all three doses of tirzepatide delivered superior A1C and weight reductions compared to the highest approved dose of semaglutide to treat type 2 diabetes," said Juan Pablo Frías, M.D., Medical Director, National Research Institute and Principal Investigator of SURPASS-2. "Newer treatment options may help people with type 2 diabetes achieve their A1C and weight loss goals. In SURPASS-2, tirzepatide delivered clinically meaningful efficacy, beyond what has been seen with an existing medicine in the established GLP-1 receptor agonist class."
SURPASS-2 was a 40-week, randomized, open-label trial comparing the efficacy and safety of tirzepatide to semaglutide as an add-on to metformin in adults with type 2 diabetes. The study randomized 1,879 participants, who had a mean duration of diabetes of 8.6 years, a baseline A1C of 8.28 percent and a baseline weight of 93.7 kg.
Across both estimandsii, all three doses of tirzepatide (5 mg, 10 mg and 15 mg) delivered superior A1C and body weight reductions compared to semaglutide. Of the participants receiving tirzepatide 15 mg, 92 percent achieved an A1C of less than 7 percent, the American Diabetes Association's recommended target for people with diabetes, compared to 81 percent of those taking semaglutide. Specifically, results showed:
In the treatment-regimen estimandiii, all three doses of tirzepatide delivered superior A1C and body weight reductions compared to semaglutide. Greater percentages of participants achieved an A1C of less than 7 percent across all three doses compared to semaglutide, with statistical significance met for 10 mg and 15 mg but not for 5 mg. Specifically, results showed:
- A1C reduction: -2.01% (5 mg), -2.24% (10 mg), -2.30% (15 mg), -1.86% (semaglutide)
- Weight reduction: -7.6 kg (5 mg), -9.3 kg (10 mg), -11.2 kg (15 mg), -5.7 kg (semaglutide)
- Percent of participants achieving A1C <7%: 82.0% (5 mg), 85.6% (10 mg), 86.2% (15 mg), 79.0% (semaglutide)
- Percent of participants achieving A1C <5.7%: 27.1% (5 mg), 39.8% (10 mg), 45.7% (15 mg), 18.9% (semaglutide)
Hypoglycemia less than 54 mg/dL was reported in 0.6 percent (5 mg), 0.2 percent (10 mg) and 1.7 percent (15 mg) of participants in the tirzepatide arms and in 0.4 percent of participants in the semaglutide arm.
The most commonly reported adverse events across all treatment arms were gastrointestinal-related, including nausea (17.4 percent [5 mg], 19.2 percent [10 mg], 22.1 percent [15 mg], 17.9 percent [semaglutide]), diarrhea (13.2 percent [5 mg], 16.4 percent [10 mg], 13.8 percent [15 mg], 11.5 percent [semaglutide]) and vomiting (5.7 percent [5 mg], 8.5 percent [10 mg], 9.8 percent [15 mg], 8.3 percent [semaglutide]). Treatment discontinuation rates due to adverse events were 5.1 percent (5 mg), 7.7 percent (10 mg), 7.9 percent (15 mg) and 3.8 percent (semaglutide).
"Nearly nine in 10 people with type 2 diabetes in the U.S. are overweight or have obesity, and we are committed to providing solutions that not only lead to meaningful A1C reductions but also significant weight loss to help meet their needs," said Mike Mason, president, Lilly Diabetes. "These striking head-to-head results surpassed our expectations, supporting our belief in the value of all three doses of tirzepatide as potential new treatment options for people living with type 2 diabetes.
"Lilly is extremely grateful to the patients and investigators who participated in SURPASS-2, particularly during the COVID-19 pandemic," Mason added. "Their commitment to the diabetes community is immeasurable."
The complete SURPASS-2 study data have not yet been evaluated but will be presented at the American Diabetes Association's® 81st Scientific Sessions® and published in a peer-reviewed publication in 2021.
About tirzepatide
Tirzepatide is a once-weekly dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist that integrates the actions of both incretins into a single novel molecule. GIP is a hormone that may complement the effects of GLP-1 receptor agonists. In preclinical models, GIP has been shown to decrease food intake and increase energy expenditure therefore resulting in weight reductions, and when combined with a GLP-1 receptor agonist, may result in greater effects on glucose and body weight. Tirzepatide is in phase 3 development for blood glucose management in adults with type 2 diabetes and for chronic weight management. It is also being studied as a potential treatment for non-alcoholic steatohepatitis (NASH).
About SURPASS-2 and the SURPASS clinical trial program
SURPASS-2 (NCT03987919) is a 40-week, multi-center, randomized, parallel, open-label trial comparing the efficacy and safety of tirzepatide 5 mg, 10 mg and 15 mg to semaglutide in adults with type 2 diabetes inadequately controlled with ≥1500 mg/day metformin alone. The trial randomized 1,879 study participants across the U.S., Argentina, Australia, Brazil, Canada, Israel, Mexico and the UK in a 1:1:1:1 ratio to receive either tirzepatide 5 mg, 10 mg or 15 mg or semaglutide 1 mg. The primary objective of SURPASS-2 was to demonstrate that the two higher doses of tirzepatide (10 mg and/or 15 mg) led to non-inferior A1C reductions from baseline compared to semaglutide after 40 weeks in people with type 2 diabetes. Key secondary objectives included non-inferior A1C reductions from baseline for tirzepatide 5 mg; superior A1C and body weight reductions from baseline and greater percentages of participants achieving an A1C less than 7 percent across all three tirzepatide doses; and greater percentages of participants achieving an A1C less than 5.7 percent for tirzepatide 10 mg and 15 mg compared to semaglutide. Additional secondary endpoints not controlled for type 1 error included percentage of participants achieving an A1C less than 5.7 percent for tirzepatide 5 mg compared to semaglutide. Study participants had a mean A1C between 7 percent and 10.5 percent and a BMI greater than or equal to 25 kg/m2. All participants in the tirzepatide treatment arms started the study at a dose of tirzepatide 2.5 mg once weekly and then increased the dose in a step-wise approach at four-week intervals to their final randomized maintenance dose of 5 mg (via a 2.5 mg step), 10 mg (via steps at 2.5 mg, 5 mg and 7.5 mg) or 15 mg (via steps at 2.5 mg, 5 mg, 7.5 mg, 10 mg and 12.5 mg). Participants in the semaglutide treatment arm started the study at a dose of semaglutide 0.25 mg once weekly for four weeks, then increased the dose to 0.5 mg for four weeks and then reached the final dose of 1 mg.
The SURPASS phase 3 global clinical development program for tirzepatide has enrolled more than 13,000 people with type 2 diabetes across 10 clinical trials, five of which are global registration studies. The program began in late 2018 with full results from the registration studies anticipated in 2021.
About Diabetes
Approximately 34 million Americans1 (just over 1 in 10) and an estimated 463 million adults worldwide2 have diabetes. Type 2 diabetes is the most common type internationally, accounting for an estimated 90 to 95 percent of all diabetes cases in the United States alone1. Diabetes is a chronic disease that occurs when the body does not properly produce or use the hormone insulin.
About Lilly Diabetes
Lilly has been a global leader in diabetes care since 1923, when we introduced the world's first commercial insulin. Today we are building upon this heritage by working to meet the diverse needs of people with diabetes and those who care for them. Through research, collaboration and quality manufacturing we strive to make life better for people affected by diabetes and related conditions. We work to deliver breakthrough outcomes through innovative solutions—from medicines and technologies to support programs and more. For the latest updates, visit http://www.lillydiabetes.com/ or follow us on Twitter: @LillyDiabetes and Facebook: LillyDiabetesUS.
About Eli Lilly and Company
Lilly is a global healthcare leader that unites caring with discovery to make life better for people around the world. We were founded more than a century ago by a man committed to creating high-quality medicines that meet real needs, and today we remain true to that mission in all our work. Across the globe, Lilly employees work to discover and bring life-changing medicines to those who need them, improve the understanding and management of disease, and give back to communities through philanthropy and volunteerism. To learn more about Lilly, please visit us at lilly.com and lilly.com/newsroom. P-LLY
Lilly Cautionary Statement Regarding Forward-Looking Statements
This press release contains forward-looking statements (as that term is defined in the Private Securities Litigation Reform Act of 1995) about tirzepatide as a potential treatment for people with type 2 diabetes and the timeline for future readouts, presentations and other milestones relating to tirzepatide and its clinical trials and reflects Lilly's current belief and expectations. However, as with any pharmaceutical product, there are substantial risks and uncertainties in the process of research, development, and commercialization. Among other things, there can be no guarantee that the studies will be completed as planned, that future study results will be consistent with the results to date or that tirzepatide will receive regulatory approvals. For further discussion of these and other risks and uncertainties, see Lilly's most recent Form 10-K and Form 10-Q filings with the United States Securities and Exchange Commission. Except as required by law, Lilly undertakes no duty to update forward-looking statements to reflect events after the date of this release.
1 Centers for Disease Control and Prevention. National Diabetes Statistics Report, 2020. Atlanta, GA: Centers for Disease Control and Prevention, U.S. Dept. of Health and Human Services; 2020.
2 International Diabetes Federation. IDF Diabetes Atlas, 9th edn. Brussels, Belgium: International Diabetes Federation, 2019. Available at: http://diabetesatlas.org.
i Efficacy estimand represents efficacy prior to discontinuation of study drug or initiating rescue therapy for persistent severe hyperglycemia.
ii Treatment differences for two estimands – efficacy and treatment-regimen – were evaluated for three tirzepatide doses (5 mg, 10 mg and 15 mg) compared to semaglutide 1 mg.
iii Treatment-regimen estimand represents the efficacy irrespective of adherence to the investigational medicine or introduction of rescue therapy for persistent severe hyperglycemia.
SOURCE Eli Lilly and Company
Posted: March 2021

