Forty Seven, Inc. Announces Updated Data from Ongoing Clinical Trial of Magrolimab Showing Robust, Durable Activity in Patients with Myelodysplastic Syndrome and Acute Myeloid Leukemia
MENLO PARK, Calif., Dec. 09, 2019 (GLOBE NEWSWIRE) -- Forty Seven, Inc. (NASDAQ:FTSV), a clinical-stage, immuno-oncology company focused on developing therapies to activate macrophages in the fight against cancer, today announced the presentation of updated clinical data from its ongoing trial evaluating magrolimab in combination with azacitidine for the treatment of myelodysplastic syndrome (MDS) and acute myeloid leukemia (AML). The new results, which will be shared in an oral presentation at the 61stAmerican Society of Hematology (ASH) Annual Meeting in Orlando, Florida, show that the combination of magrolimab and azacitidine is highly active and well-tolerated in patients with MDS and AML.
“As a clinician caring for patients with MDS, I am reminded daily of the limitations of existing standard-of-care medicines, which are appropriate for a small fraction of people and leave nearly 75% receiving only supportive care or watchful waiting,” said David Sallman, M.D., H. Lee Moffitt Cancer Center and Research Institute, an investigator for the clinical trial. “The data that continue to emerge from this clinical trial are incredibly exciting, suggesting that the combination of magrolimab and azacitidine may offer the first new therapeutic regimen in over a decade, with the potential to induce meaningful and lasting responses in patients with higher-risk disease. Importantly, these results also support magrolimab’s tolerability profile, further differentiating it as a safe treatment that may be used even in more fragile, sicker, and older patients.”
“We are very encouraged by the tolerability and sustained clinical activity of the magrolimab and azacitidine combination observed thus far, which increase our confidence in the program and reinforce our commitment to advancing it as rapidly and as responsibly as possible,” said Chris Takimoto, M.D., Ph.D., F.A.C.P., Chief Medical Officer of Forty Seven. “To that end, we are pleased with our recent interactions with the U.S. Food and Drug Administration (FDA), and we appreciate the agency’s feedback as we finalized the design of our registration-enabling program. With its guidance, we have developed a protocol that allows us two distinct opportunities to achieve an accelerated approval, while remaining on track to achieve our goal of filing an initial biologics license application (BLA) in the fourth quarter of 2021. We look forward to working with our investigators, clinicians and the broader patient community as we advance magrolimab forward for people living with MDS.”
Data from the Ongoing Phase 1b Clinical Trial
Forty Seven’s Phase 1b trial, which is being funded in part by the California Institute of Regenerative Medicine (CIRM), is designed to evaluate magrolimab in combination with azacitidine in untreated patients with higher risk MDS and untreated patients with AML, who are ineligible for induction chemotherapy. All patients received a 1 mg/kg priming dose of magrolimab, coupled with intrapatient dose escalation, to mitigate on-target anemia. Patients were then treated with full doses of azacitidine and a magrolimab maintenance dose of 30 mg/kg once weekly.
As of the data cutoff of November 18, 2019, 62 patients had been treated with the combination in the Phase 1b portion of the trial, including 35 patients with MDS and 27 patients with AML.
Clinical Activity Data
As of the data cutoff, 46 patients were evaluable for response assessment, including 24 patients with untreated higher-risk MDS and 22 patients with untreated AML, who are ineligible for induction chemotherapy.
In higher-risk MDS, the overall response rate (ORR) was 92%, with 12 patients (50%) achieving a complete response (CR), eight patients (33%) achieving a marrow CR and two patients (8%) achieving hematologic improvement. Additionally, two patients (8%) achieved stable disease.
In untreated AML, the ORR was 64%, with nine patients (41%) achieving a CR, three patients (14%) achieving a CR with complete blood count recovery (CRi) and one patient (5%) achieving a morphologic leukemia-free state (MLFS). Additionally, seven patients (32%) achieved stable disease (SD) and one patient (5%) had progressive disease.
The median time to response among MDS and AML patients treated with the combination was 1.9 months.
No median duration of response or overall survival has been reached for either MDS or AML patients, with a median follow-up of 6.4 months (range 2.0 to 14.4 months) for MDS and 8.8 months (range 1.9 to 16.9 months) for AML.
Additionally, mutational analyses are ongoing to correlate subgroups with response. Seven of nine (78%) evaluable TP53 mutant AML patients achieved an objective response, with 44% achieving CR and 33% achieving CRi. TP53 mutations are often associated with a poor prognosis and patients with TP53 mutant disease are refractory to existing therapies.
Lastly, in AML patients who achieved an objective response, a significant increase in CD4 and CD8 T cell infiltration was observed in the bone marrow while on therapy, demonstrating that magrolimab and azacitidine can induce an adaptive T cell response.
Safety Data
As of the data cutoff, the combination of magrolimab and azacitidine was well-tolerated, with no evidence of increased toxicities compared to azacitidine alone. Adverse events (AEs) were consistent with prior clinical experience. No deaths were observed in the first 60 days on combination treatment and only one patient out of 62 (1.6%) discontinued treatment due to a treatment-related AE.
Clinical Development Plans for Magrolimab in MDS and AML
Forty Seven achieved alignment with the FDA on the final design of its potentially registration-enabling clinical development program for magrolimab in higher-risk MDS. Based on recent interactions under its Special Protocol Assessment (SPA), Forty Seven now plans to continue enrolling patients in its ongoing Phase 1b clinical trial with an amended protocol, and to initiate a Phase 3 randomized control trial (RCT), ENHANCE, to evaluate the combination of magrolimab and azacitidine compared to azacitidine alone. This approach provides two distinct opportunities to achieve an accelerated approval, with a potential BLA filing expected in the fourth quarter of 2021.
Forty Seven has amended the protocol for its ongoing Phase 1b clinical trial to evaluate dosing every two weeks, and remains on track to complete enrollment of approximately 90 patients in the third quarter of 2020. ENHANCE, which will initiate in the first half of 2020, will enroll approximately 180 patients, randomized 1:1 to receive magrolimab and azacitidine or azacitidine alone. For both studies, the primary endpoint will be CR rate and duration of response, and either study could serve as the basis for an accelerated approval. ENHANCE will have an adaptive design, such that it can be extended to include additional patients evaluated for a longer period of time for overall survival benefit in support of a potential full approval based on maturing data in the ongoing Phase 1b trial. Additionally, given the decision to design ENHANCE as an RCT with standard, well-validated endpoints, the SPA process is no longer required.
Given the encouraging data observed in patients with TP53 mutant AML and the significant unmet medical need in this population, Forty Seven will continue to enroll additional patients with TP53 mutant disease in the ongoing Phase 1b trial, with potential future regulatory interactions to be considered pending analysis of the maturing data.
About Magrolimab
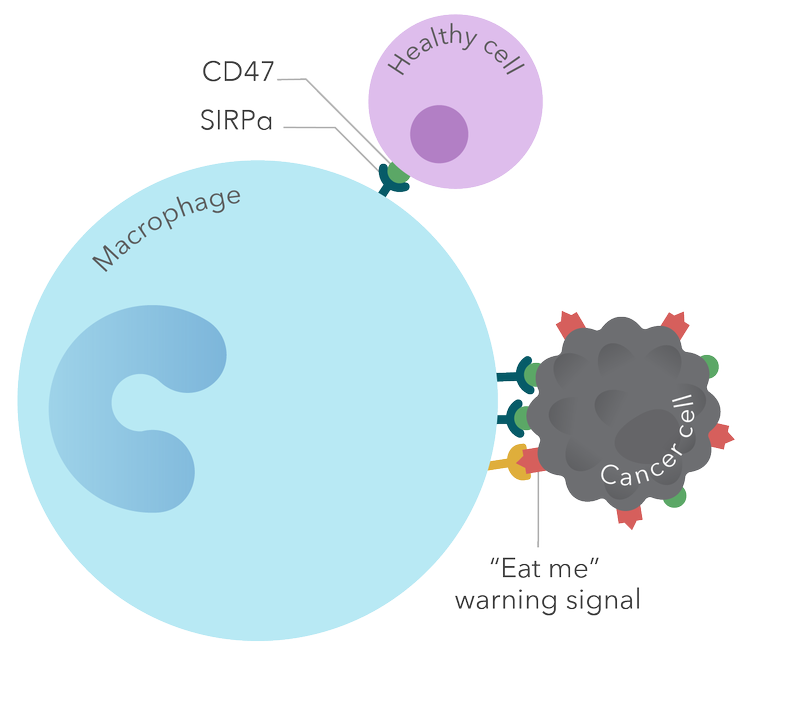
Magrolimab is a monoclonal antibody against CD47 that is designed to interfere with recognition of CD47 by the SIRPα receptor on macrophages, thus blocking the "don't eat me" signal used by cancer cells to avoid being ingested by macrophages. Forty Seven is initially developing magrolimab, an investigational medicine, for the treatment of patients with MDS, AML, non-Hodgkin’s lymphoma, ovarian cancer and colorectal carcinoma. Magrolimab has been granted Fast Track designation by the FDA for the treatment of MDS and AML, and for the treatment of relapsed or refractory diffuse large B-cell lymphoma (DLBCL) and follicular lymphoma, two forms of B-cell non-Hodgkin's lymphoma. Magrolimab has also been granted Orphan Drug designation by the FDA and European Medicines Agency for the treatment of AML. (Article from : www.drugs.com)

